How deliberately infecting volunteers with COVID-19 could help accelerate vaccine development
Josh Morrison, Co-founder and Executive Director of 1Day Sooner, an organisation that advocates on behalf of challenge trial volunteers, explains why.
- 5 November 2020
- 5 min read
- by Gavi Staff
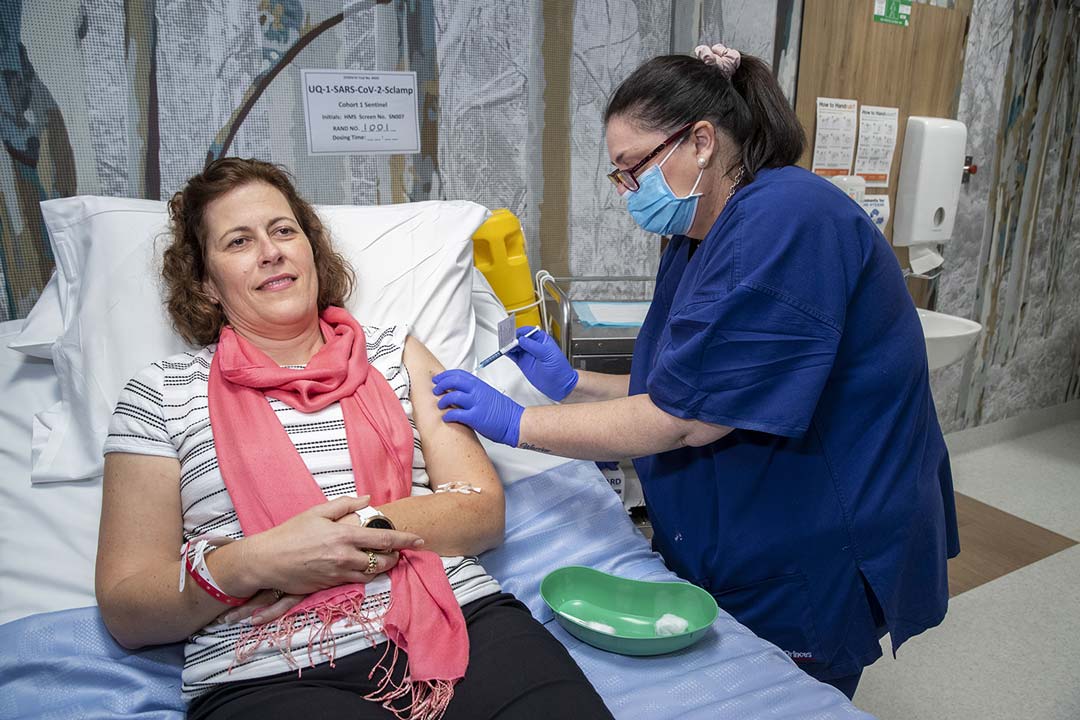
The COVID-19 vaccine race is both a marathon and a sprint. While there are promising signs that the first sprint may be over and that an effective vaccine may start to become available in some countries by the end of this year, the first vaccines are rarely the best ones. If the goal is to vaccinate the world, a number of vaccines will need to be proven effective beyond the first wave in order to reach the 16 billion doses (since most potential vaccines require a booster) that will likely be needed to vaccinate everyone. Research into SARS-CoV-2 vaccines and treatments will need to continue at least throughout the next year, and human challenge studies will be a crucial tool to help scientists in the marathon effort of exploring and ultimately eradicating the disease.
A “challenge trial” (otherwise known as a controlled human infection study) is a way of studying a disease by deliberately exposing healthy volunteers in a controlled setting. They have been key to our understanding of a number of diseases, like influenza, respiratory syncytial virus and the common cold, and have played an important role in the development of vaccines for malaria, typhoid and cholera among others. As Oxford vaccinologists Adrian Hill and Alexander Douglas recently wrote in the Nature Immunology Reviews, challenge studies “allow for the time, dose, route and strain of the infectious exposure to be controlled by the investigator.” This makes them invaluable for answering a number of critical questions about SARS-CoV-2: How long does immunity last? What happens in the early immune response upon exposure? How can we predict if a vaccine provokes a response that correlates with effective protection?
Without challenge studies, which allow scientists to observe the course of infection from its beginning, these questions become far more difficult to answer.
Of course, deliberately exposing people to a deadly disease requires great care and caution. We still don’t fully understand the risks of long-term COVID. A recent pre-print found that 1 in 40 British COVID patients had symptoms after 12 weeks. (Age, health, and severity of disease correlate with long-term symptoms.) Plus, COVID-19 can be deadly even for young, healthy people. Still, the known risks of death are meaningfully lower than other risks we commonly allow people to take, such as living kidney donation or childbirth in the United States. And there are people who are willing to volunteer. Nearly 40,000 people have expressed interest in participating in a challenge trial with 1Day Sooner, a non-profit I co-founded to advocate for these volunteers. These people understand the risks, but believe that the benefits far outweigh them.
Have you read?
In addition to helping us answer basic questions about SARS-CoV-2, challenge studies also allow scientists to test vaccines and treatments more rapidly and with greater precision. Pfizer’s current Phase 3 study requires a minimum of 31 cases of infection to start assessing whether a vaccine protects more people in the treatment group than were infected in the control. Yet, out of 30,000 people vaccinated by the end of the summer, it failed to reach 31 infections before November. A challenge study could reliably assess that question for vaccines and treatments within weeks rather than months, and with a few dozen volunteers instead of tens of thousands.
Similarly, they could help us select the best vaccines to test. For example, according to the World Health Organization, there are 45 vaccine candidates in clinical evaluation. They cannot all receive massive 30,000 person Phase 3 trials. Challenge studies can help “down-select” between promising vaccine candidates to prioritise the ones that are likely to work well, and justify the hundreds of millions of dollars of investment needed for a Phase 3 trial.
This would have particular value for the developing world. Adar Poonawalla, Chief Executive of the Serum Institute of India, recently predicted that, due to limited manufacturing capacity, there won’t be enough vaccine doses for worldwide distribution until the end of 2024. Clearly, we need to develop as many safe and effective vaccines as possible. Moreover, many of the first generation of vaccines will be difficult to deliver to low-income countries because they may require booster shots and storage at ultra-cold temperatures, well below what traditional cold-chain equipment is capable of. Developing vaccines that do not require cold storage or a booster shot will be invaluable to ending COVID-19 throughout the world.
To be clear, challenge studies are not a perfect substitute for conventional Phase 3 trials. They study artificial infection in young, healthy people, and safety studies in large groups would still need to be conducted. COVID-19 challenge studies are not a silver bullet, but they are an essential way of advancing our understanding of this disease and getting vaccines to those who need them as soon as possible. Thousands of young healthy volunteers have stepped forward to take part in these trials and the UK recently announced a plan to begin challenge studies in January 2021. Others should follow suit.