Are African countries ready for the COVID-19 vaccine?
Preparing African countries for COVID-19 vaccines will require thoughtful planning and unprecedented coordination across a wide-range of stakeholders.
- 10 December 2020
- 5 min read
- by Gavi Staff
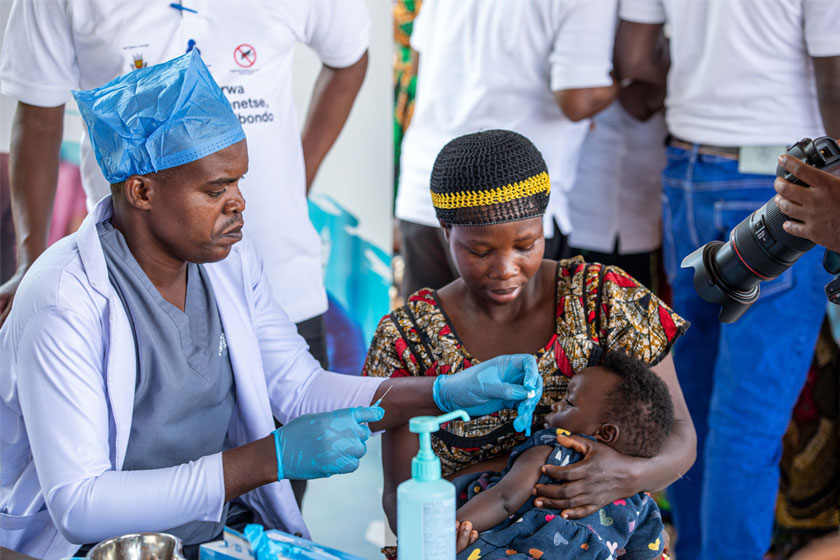
The COVID-19 pandemic has led to the loss of thousands of lives and disrupted millions more on the African continent. As scientists race to find, manufacture and deliver a safe and effective vaccine, country preparedness for an eventual vaccine rollout is essential alongside commitments to ensure equitable access to them. A steep misinformation curve is also likely to affect community trust and confidence in immunisation. All of this must be addressed if African governments are going to succeed in vaccinating their populations against COVID-19.
Because ultimately, vaccines alone don’t save lives, vaccination does.
Dr. Matshidiso Moeti, WHO Regional Director for Africa, in her call for equitable access to vaccines, noted that African countries are too often at the back of the queue for new technologies and public health interventions, including vaccines. The continent’s leadership should remain engaged and involved in the eventual COVID-19 vaccine rollout, ensuring wide distribution to meet the population’s needs.
It is commendable to see global leaders come together under the WHO-led Access to COVID-19 Tools Accelerator, which includes the COVID-19 Global Vaccine Access Facility (COVAX). African leaders have equally expressed their commitment to securing an effective vaccine for their populations through the Consortium for COVID-19 Vaccine Clinical Trial (CONCVACT) led by the African Union.
In partnership with PATH and the Gavi CSO Steering Committee, Dalberg Advisors and Amref Health Africa hosted the Africa Dialogues webinar on African country readiness for a COVID-19 vaccine. Panelists opined on critical issues that could impede African countries’ preparedness for equitable delivery of the highly anticipated vaccines, including funding gaps, weak health systems, poor supply chain infrastructure and undefined eligibility and prioritisation criteria to ensure that the most vulnerable populations receive access as soon as possible.
Watch the Africa Dialogues webinar
The panelists proposed the following priority actions for governments to help Africa prepare for COVID-19 vaccines:
- ENGAGE communities to address mistrust, minimise vaccine hesitancy and to better inform key opinion leaders and the general public on the need for the vaccine and the benefits thereof, for increased uptake.
- REMOVE barriers to regulation and approval of COVID-19 vaccines across Africa, by accelerating harmonised regulatory processes and fast-track country authorisation of safe and effective COVID-19 vaccines.
- LEVERAGE African countries’ participation in the COVAX Facility to strengthen pooled bargaining power and ensure Africa can access a sufficient share of the global vaccine supply.
- ALLOCATE funding to secure COVID-19 vaccines and strengthen the capacity of immunisation service delivery systems including the supply chain, cold chain requirements for the vaccine and demand generation activities.
- PROMOTE regional manufacturing capacity for vaccines to support the continent’s demand for COVID-19 vaccines by enhancing inter-country trade.
- MAINTAIN, restore and strengthen routine immunisation services and recovery from COVID-19 related disruptions.
To achieve the goal of vaccinating at least 60% of the population, Africa will need about 1.5 billion vaccine doses which at current estimates could cost between US$ 8 billion and US$ 16 billion, with additional mark-up costs of 20-30% for vaccination programme delivery. What does this mean for African countries now and for future pandemics?
Leadership from the Africa Centres for Disease Control and Prevention (Africa CDC) has been paramount in coordinating Africa’s COVID-19 response. In its recently published COVID-19 Vaccine Development and Access Strategy, the first key objective is to accelerate Africa’s involvement in research and development. According to the African Academy of Sciences, only 2% of clinical trials conducted worldwide occur in Africa. Investments in research and development will ultimately lead to increased efficacy of vaccines for populations across the continent.
The time to establish structures suitable for effective immunisation is now. In addition to involvement in clinical trials countries should prioritise funding for immunisation and follow established guidelines for country readiness and delivery. For example, plans must be in place for where the vaccine will be administered and by whom, along with protocols for safe and efficient vaccination processes in clinics and in mass vaccination settings that are COVID-19 safe and have optimal record-keeping, reporting systems, eligibility criteria and vaccine schedules.
Because ultimately, vaccines alone don’t save lives, vaccination does.