Lab-grown blood used in transfusion for first time – here are three other ways that making organs could change healthcare
Lab-grown technologies may help change the way certain human diseases are studied and treated.
- 13 December 2022
- 5 min read
- by The Conversation
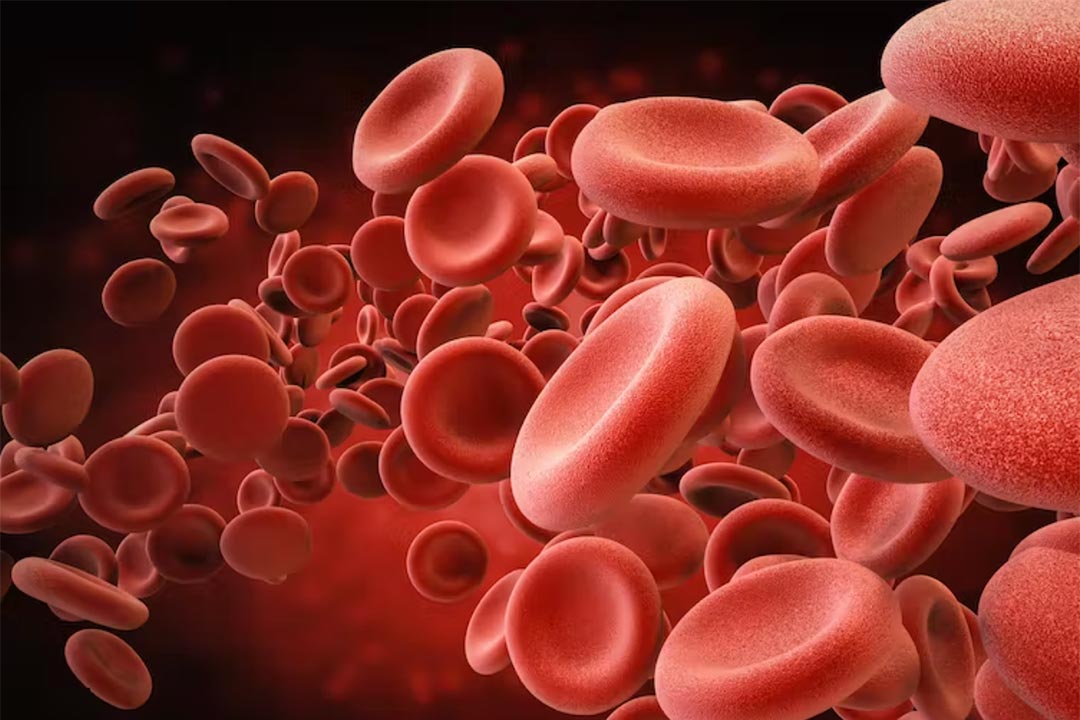
Human volunteers have received the first-ever transfusion of lab grown red blood cells in a groundbreaking scientific first. Volunteers received about 5ml-10ml of blood (around 1-2 teaspoons), which contained around 15 billion red blood cells. So far, volunteers who received the lab-grown blood cells are reported to be in good health.
To grow these red blood cells, researchers first extracted stem cells from a blood sample. They then directed these stem cells to grow into red blood cells in the laboratory. It’s hoped that these lab-grown blood cells may be used in the treatment of disorders that affect red blood cells – such as sickle cell anaemia and thalassemia.
While this is the first time blood has ever been grown in the lab, it certainly isn’t the first lab-grown innovation. Scientists have used laboratory-based models for years to investigate everything from organ and tissue function, to understand disease processes and to test new treatments.
Here are some of the other most promising lab-grown innovations to date.
Organoids
In recent years organoids, derived from various types of stem cells, have become more common in research. Organoids closely match the structure and function of full-size human organs. This allows researchers to study how many different diseases or viruses may affect human health. They also allow researchers to better understand stem cells (which they grown from), which can become almost any cell in the body.
For instance, organoid brains have helped scientists understand the devastating effect of Zika virus on brain development. They’re also playing a wider key role in understanding different neurological conditions, such as ALS. Organoid brains are extremely advantageous because they allow researchers to observe, in real-time, any changes that may occur in the brain as they happen – something not possible with humans. This will help us better know when and how to treat certain diseases.
Organoid hearts have also been successfully grown in the lab. After a week of development, they’re functionally equivalent to the heart of a 25-day-old embryo, able to beat between 60-100 times per minute. Recent advances have also enabled heart cells to be grown from stem cells, paving the way to be able to grow larger and more efficient cardiac organs.
The cells from these organoids have also been shown to improve the performance of damaged heart muscle when given to animals. Similar work shows that they are also providing valuable information in what happens in heart attack, heart failure and a number of genetic heart conditions.
Have you read?
Organoid lungs are also proving useful in understanding COVID-19 and testing new therapies, alongside helping scientists understand other diseases, such as chronic obstructive pulmonary disease, asthma and certain types of pulmonary fibrosis. These kind of human lung organoids take approximately eight weeks to grow in the lab.
Fully-grown organs
Scientists have also been able to successfully grow fully-functioning organs in the lab.
For example, a small number of people are actually walking around with fully-grown, functioning bladders made from their own cells. These are produced in a lab by growing the patient’s own bladder cells and then 3D-printing them into the correct shape. It’s then surgically implanted into the patient. This process takes around two months to complete. But since these are grown from the patient’s own cells, it removes the risk of the body rejecting the new organ.
Lab-grown vaginas are another example. These are usually grown and implanted into women who are born without a vagina – such as those suffering from a rare disease called Mayer-Rokitansky-Küster-Hauser Syndrome. The process is somewhat similar as with bladders, whereby cells from their external genitalia are used to grow the vagina in a bio-reactor.
One of the biggest challenges that researchers now face is developing lab-grown skin. Skin is the largest organ in the body and is composed of three distinct layers. But finding a way of integrating these layers in the same way as they are in the body is proving difficult for scientists.
While skin cells have been successfully grown in labs for many years, it still takes between 2-3 weeks to grow enough skin cells to fill the area they’re being grafted onto. However, co-culturing them with cells from the underlying dermis (one of the skin’s three layers) has shown to improve yield and performance of lab-grown skin cells. Another challenge researchers face is growing skin which contains elements such as sweat glands and hairs. These are essential as they help the skin perform one of its many important functions: regulating body temperature.
Ears
Scientists have successfully grown and 3D-printed a human ear using a patient’s cartilage cells.
It’s hoped that this development will help in treating people born with microtia or anotia – a birth defect that causes the ear not to form properly, or not form at all. The condition effects approximately one in every 2,000-10,000 births. But while the external ear improves appearance, there’s still the challenge of overcoming hearing loss due to the initial absence of the ear, which can also impact speech.
Almost 6,800 people are waiting for an organ transplant in the UK and more than 15 times that in the US. Advances in lab grown organs may provide an alternative for those waiting for transplants, and can be produced from a patient’s own cells.
Future innovations are looking towards other lab grown organs such as kidneys and livers, as these represent a large portion of the organs that people waiting for transplants need.![]()
Authors
Adam Taylor, Professor and Director of the Clinical Anatomy Learning Centre, Lancaster University

This article is republished from The Conversation under a Creative Commons license. Read the original article.
Disclosure statement
Adam Taylor is affiliated with the Anatomical Society.
Partners
Lancaster University provides funding as a founding partner of The Conversation UK.







