Will coronavirus herald a new era in vaccine innovation?
A vaccine against the novel coronavirus is under urgent development as the number of people infected with COVID-19 increases, a process which could also result in a new range of highly innovative vaccine technologies that ushers in a new era in the way we fight infectious disease.
- 10 March 2020
- 6 min read
- by Ciara McCarthy

As this novel coronavirus continues to spread, and the number of people infected with COVID-19 keeps rising, the race is on to develop a vaccine. But the time it takes to carry out clinical trials, to ensure that a vaccine is both safe and effective, means this won’t happen overnight.
However, with at least 35 coronavirus vaccines now being developed, even compared to recent pandemics, we have new tools available that could help us reach that goal in record time.
This has implications not just for the rapid availability of a COVID-19 vaccine for the current outbreak. The sheer variety of the novel types of vaccines being investigated and the approaches they use, could serve us well for future outbreaks of other novel diseases. In the process we could finally see a range of highly innovative vaccine technologies take off that are radically different from traditional approaches, ushering in a new era in the way we fight infectious disease.
Vaccine reboot
One way in which vaccine development can be sped up is to build on work carried out on existing vaccines, which may have already undergone some of the clinical trials needed. For example, in 2010 a vaccine for H1N1 pandemic influenza became available for widespread use within only six months of the identification of the new influenza strain because the pandemic vaccine was similar to vaccines used to protect against previous seasonal influenza strains.
In addition, during the two recent major coronavirus outbreaks — the first in 2003 with Severe Acute Respiratory Syndrome (SARS) and then in 2012 the Middle East Respiratory Syndrome (MERS) — significant progress was made in developing vaccines against these infections. Researchers now are leveraging some of this work to accelerate progress towards a COVID-19 vaccine.
This doesn’t always have to be based on vaccines for related diseases. For example, the Ebola vaccine, which has been used to help prevent the recent epidemic spiralling out of control in the Democratic Republic of the Congo, uses vaccine technology originally created for the development of an HIV vaccine. Similarly, the Pasteur Institute is now using an approach that co-opts an existing measles vaccine as a backbone, with COVID-19 antigens added to it.
The advantage of using existing “platforms” like this is that the time it takes to carry out clinical trials can be massively reduced because new vaccines can potentially be shown to be as safe and effective as existing similar vaccines instead of starting from scratch.
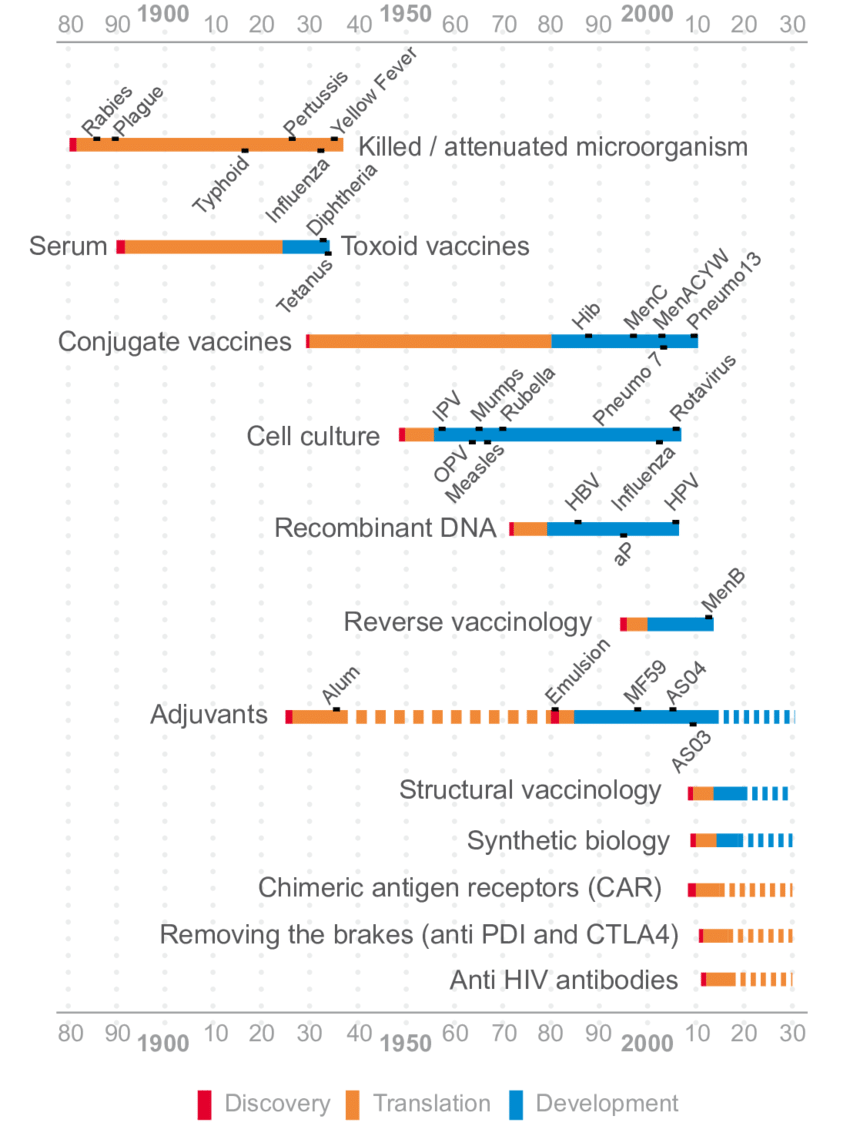
Source: Stevens, Hilde & Debackere, Koenraad & Goldman, Michel & Mahoney, Richard & Stevens, Philip & Huys, Isabelle. (2017). Vaccines: Accelerating Innovation and Access Global Challenges Report.
DNA vaccines
In recent years, we have also seen the development of completely novel vaccine technologies that could dramatically speed up vaccine development, even against infectious diseases that humanity has never seen before.
Traditionally, most vaccines consist of a killed or attenuated form of the disease-causing virus or bacteria. When the vaccine has been administered, our body responds by producing antibodies against one or more antigens, such as proteins or carbohydrates, from that specific germ. This means that if we become infected in the future, our immune system is already primed to rapidly recognise and tackle the infection.
Newer DNA-based vaccines take a different approach. For coronavirus, rather than being made up of the coronavirus itself, they involve injecting a fragment of coronavirus DNA, containing instructions to build the antigens found on the surface of the coronavirus. Our cells use these instructions to assemble the antigens themselves. These are essentially just fragments of the virus which by themselves don't pose the same threat as the virus. But our immune system still recognises them as foreign and produces antibodies capable of recognising the complete virus in future. Rather than waiting to obtain a sample of the virus and grow it in the lab before injecting killed or attenuated versions into the body, researchers are essentially outsourcing to our own cells the job of producing the antigens that teach our bodies how to recognise germs.
Other groups are developing mRNA vaccines that work in much the same way. Both methods have a number of potential advantages over traditional vaccines, but a significant one is the speed at which a candidate vaccine can be created. Finding a way to produce a suitable antigen on which to base a vaccine can take considerable time. But with DNA and mRNA vaccines you just need the genome. So in January when rapid genetic sequencing of the coronavirus was published by Chinese scientists it was possible to develop a vaccine within just days.
Another big advantage of approaches like these is their adaptability. They are sometimes called “plug-and-play” vaccines, as in theory DNA or RNA from a huge number of germs can be “plugged” into the same vaccine platform. When it comes to developing vaccines against novel infectious diseases, this really could be a gamechanger.
Molecular clamps
A variant on a more traditional protein-based vaccine approach, which involves injecting just proteins, attached to nothing, directly into the body, is also being pursued for coronavirus vaccines.
These proteins are copies of those found on the surface of the coronavirus. By themselves, these coronavirus proteins would normally be unstable, and rapidly lose their shape after injection, making it difficult for the immune system to produce antibodies specific to the virus.
But researchers at the University of Queensland, in Australia, have developed a “molecular clamp” which helps ensure the viral proteins retain their shape, enabling the immune system to mount a much stronger immune response. Human papillomavirus vaccines have previously shown the value of being able to base a vaccine on free floating proteins, so this general approach has a solid track record.
All of these approaches are moving at an unprecedented pace. This is in part due to support from the Coalition for Epidemic Preparedness Innovations, or CEPI, which was set up in 2017 to accelerate development of vaccines against emerging infections with epidemic potential. If all goes extremely well, an investigational vaccine could be available for widespread distribution and use within 12-18 months. For context, this is a process that has often taken well over a decade for previous vaccines.
Gavi is now looking at the potential role it can play when a vaccine or vaccines become available, to ensure that everyone who needs it has access to it, regardless of their wealth. With the current outbreak, the hope is that the outbreak will be swiftly brought to an end through non-pharmaceutical interventions.
The development of a vaccine offers an insurance policy in case this does not happen, but hopefully that insurance policy will not be required. However, even in that case, the work carried out on coronavirus vaccines today will almost certainly help us be better prepared for the next outbreak, whatever form that takes.
Updated 12 March 2020