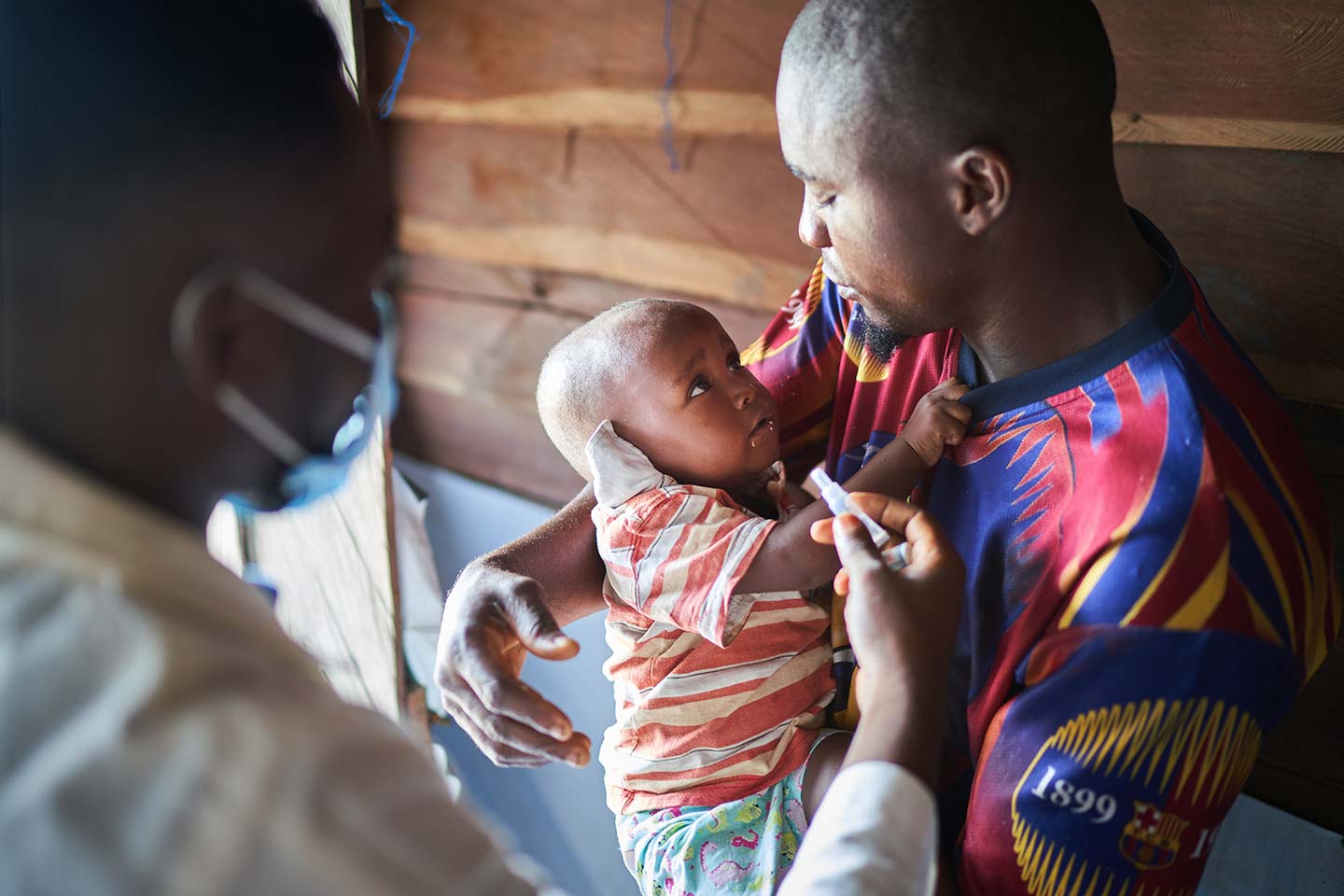
What you need to know about COVID-19 vaccine approvals in Nigeria and South Africa
Before any COVID-19 vaccines can be delivered to the public, their use must be approved by national regulatory authorities. We look into the process before vaccines would be approved for distribution and use in Nigeria and South Africa.
- 22 December 2020
- 4 min read
- by Tetsekela Anyiam-Osigwe
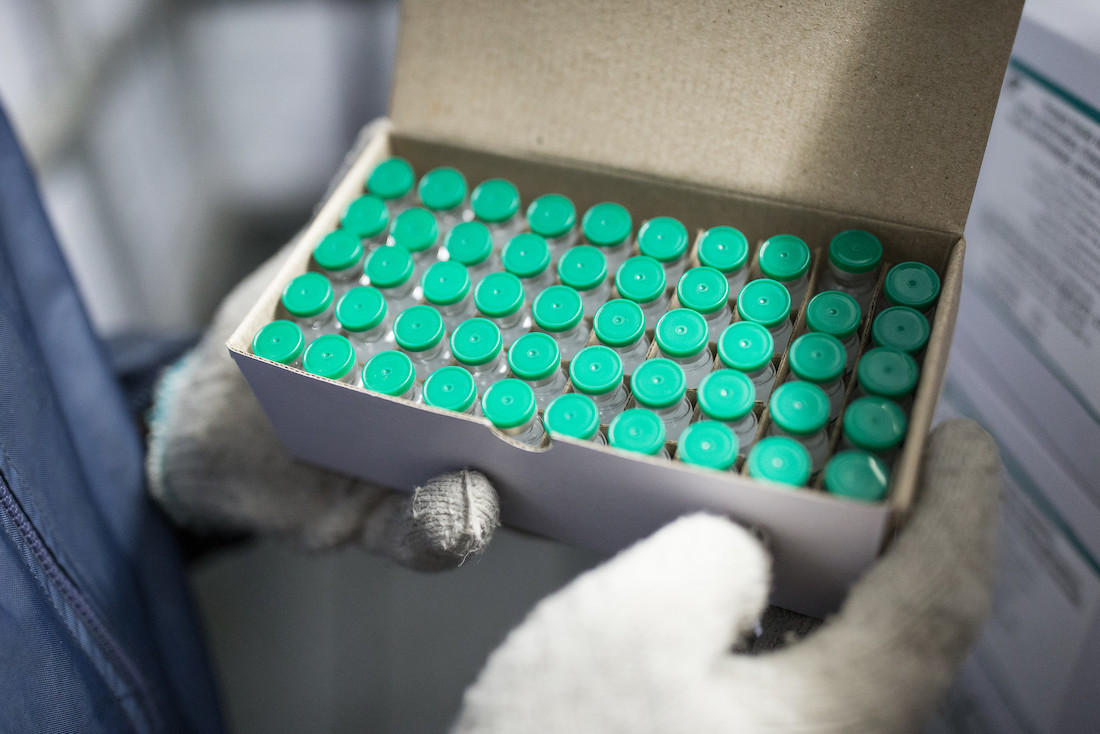
Normally, vaccines would take several months and, sometimes, even years to be approved by regulatory authorities. However, due to the urgent need for COVID-19 vaccines to end the acute phase of the pandemic, normal regulatory processes are being adapted to move more quickly. South Africa and Nigeria have tailored their own review processes in order to expedite COVID-19 vaccine approvals, while still ensuring that the highest safety standards are met.
While Nigeria expects its first approval at the end of the first quarter or the beginning of the second quarter of 2021, South Africa’s first approval might come earlier.
Who would approve the vaccine?
The Nigerian National Agency for Food and Drug Administration and Control (NAFDAC) and the South African Health Products Regulatory Authority (SAHPRA) have oversight of regulatory checks and approvals for public delivery and the use of medicines and other critical health technologies, including vaccines, within their countries. All vaccines will need to be registered with these agencies, whose job it is to ensure that they meet the necessary standards for safety, quality and efficacy.
How are COVID-19 vaccine approvals being fast-tracked?
Given the extraordinary circumstances and devastating impacts of the COVID-19 pandemic, vaccines have to reach people across the country, as quickly as possible, and work safely and effectively. Both NAFDAC and SAHPRA will be prioritising COVID-19 vaccine applications and have procedures set in place to accelerate the approval process:
Procedure 1: Prequalification
NAFDAC and SAHPRA will be leveraging the World Health Organization’s (WHO) long-established prequalification procedures. Such leveraging facilitates the efficient registration of WHO-approved vaccines in low- and middle-income countries. Nigeria and South Africa will also be relying on decisions by major national and regional regulators, including the US Food & Drug Administration (FDA) and European Medicines Agency (EMA), which have committed to maximising transparency surrounding their approvals of COVID-19 vaccines. This means that vaccines that have already gained approval from these external regulators will qualify for both NAFDAC and SAHPRA’s fast-track domestic review process for regulatory approval and licensing. Such a fast-track approach is likely to see reviews completed in just 15 days rather than several months, according to NAFDAC’s Director-General, Mojisola Adeyeye.
Have you read?
Procedure 2: Safety monitoring
Information from vaccine manufacturers, including results from clinical trials, will be further evaluated by NAFDAC and SAHPRA as an additional safety mechanism to ensure that vaccines satisfy all regulator-specific standards for use in Nigeria and South Africa. In addition to data robustness checks, NAFDAC plans to assess risk management plans and vaccine manufacturing practices. Meanwhile, SAHPRA’s spokesperson, Yuven Gounden, has said that the agency has already established a specialised COVID-19 Vaccine Registration Working Group to prioritise and expedite reviews of COVID-19 vaccines in a way that ensures that all of SAHPRA’s requirements of quality, safety and efficacy are satisfied.
When should the first approvals be expected?
While Nigeria expects its first approval at the end of the first quarter or the beginning of the second quarter of 2021, South Africa’s first approval might come earlier. SAHPRA received its first vaccine application on 10 December from US-based Johnson & Johnson and has already begun a rolling review process that evaluates vaccine candidate data as it becomes available. However, Johnson & Johnson’s vaccine, one of at least four vaccines being trialled in South Africa, will first need to provide satisfactory results in its phase 3 trials before any approval is likely. Both South Africa and Nigeria are also part of the Gavi COVAX AMC Facility and are on course to receive their first vaccines from COVAX next year.