Could a rapid neutralising antibody test free up doses of COVID-19 vaccines for low- and middle-income countries?
By identifying people with high levels of neutralising antibodies after a first vaccine dose, rapid tests could potentially identify those who don’t need a booster dose.
- 29 June 2021
- 4 min read
- by Linda Geddes
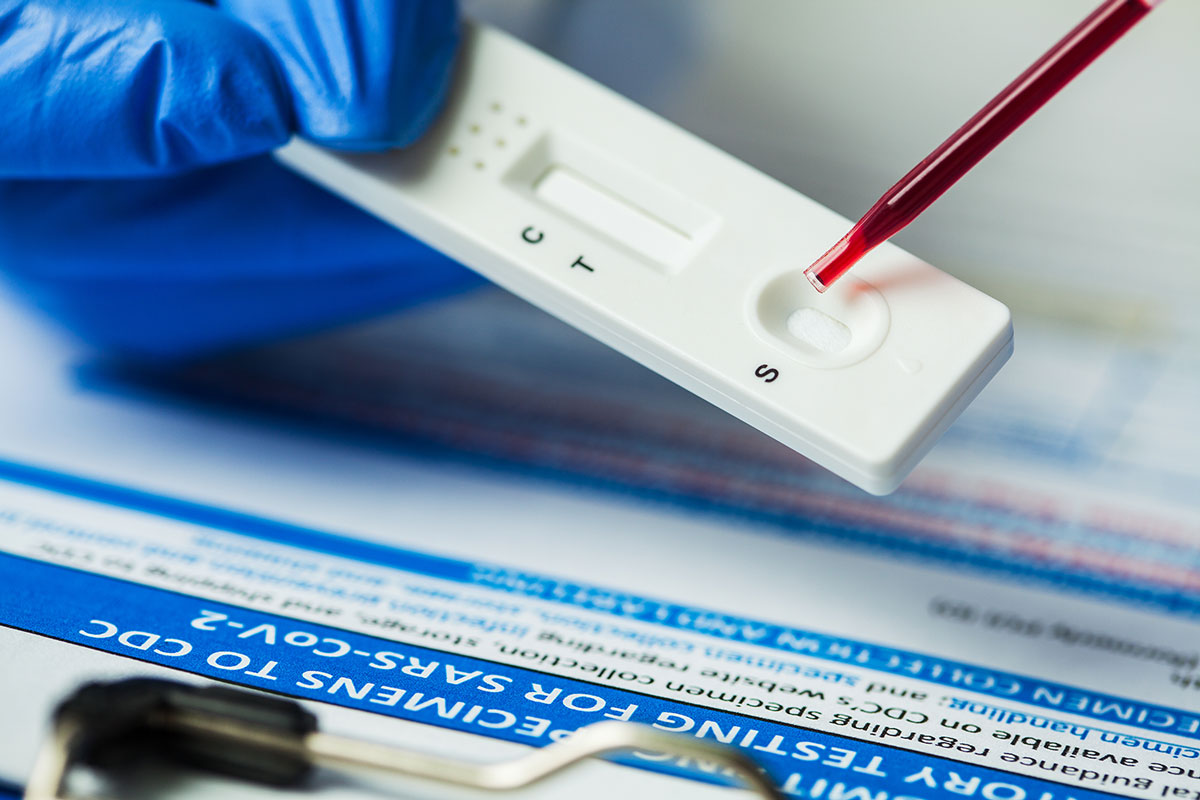
Most of the COVID-19 vaccines that have been authorised so far require two doses for maximum efficacy. However, several studies have suggested that, for people who have already had COVID-19, a first dose may be equivalent to a second dose in terms of the protective immunity it provides.
In theory, this might allow those precious second vaccine doses to be diverted towards individuals in greater need, such as frontline health workers in low- and middle-income countries who haven’t yet received a first dose. The problem is that not everyone who has experienced COVID-19 has the same level of immunity, meaning some people with a past infection may need not only a first vaccine dose to be protected, but a second dose as well.
A neutralising antibody test is more informative about your immunity to the virus.
Neutralising antibodies
Enter “rapid neutralising antibody tests”. Unlike first generation antibody tests, which detect all antibodies that bind to the virus that causes COVID-19 – including some which have little influence on whether the body has the ability to defeat the infection – a neutralising antibody test only detects those that block the SARS-CoV-2 virus from infiltrating cells and replicating.
Neutralising antibodies are sometimes missing in people who have had COVID-19, meaning that they can be reinfected, but vaccines are better at triggering this type of lasting protective immunity.
Although either type of test can detect if you’ve previously been infected with SARS-CoV-2, a neutralising antibody test is more informative about your immunity to the virus.
Such tests could also be used to monitor people’s individual responses to vaccines, potentially identifying those who need additional booster doses, or as a research tool to identify whether certain groups of individuals, such as older or vulnerable people, need additional boosters, and if so, when.
The presence of neutralising antibodies could even be used as a form of social passport, enabling those with a positive test to enjoy greater freedoms. For instance, Hong Kong recently announced that fully vaccinated travellers who also test positive for neutralising antibodies could undergo a seven-day quarantine, rather than the usual 21 days.
Antibody test
Like many general COVID-19 antibody tests, neutralising antibody tests involve testing a small blood sample. Some of these tests are designed for use in a laboratory, while others are Rapid Diagnostic Tests. These RDTs involve placing blood onto an absorbent pad at one end of a small plastic cartridge and waiting 20-30 minutes for the result to become visible within a separate reading window in the cartridge.
Have you read?
Some of these tests have received emergency use authorisation in individual countries, such as the US, but the World Health Organization has not yet approved any of them.
A critical question is how reliable they are. If a test were to be used to decide who only needed one vaccine dose, or if certain individuals could be exempt from COVID-19 restrictions, it would need to be highly accurate. The first generation of general COVID-19 antibody tests were initially hailed as the key to getting back to “normality,” but subsequent studies indicated that a positive antibody test didn’t necessarily imply immunity.
Further validation
The US Food and Drug Administration (FDA), which authorised its first neutralising antibody test in November 2020, cautioned against people using the results from these tests (or any antibody test) as an indication that they could stop taking steps to protect themselves and others, such as stopping social distancing, discontinuing wearing masks or returning to work.
“There are still many unknowns about what the presence of SARS-CoV-2 antibodies may tell us about potential immunity, but today’s authorisation gives us another tool to evaluate those antibodies as we continue to research and study this virus,” said Tim Stenzel, Director of the Office of In Vitro Diagnostics and Radiological Health in the FDA’s Center for Devices and Radiological Health. “Patients should not interpret results as telling them they are immune, or have any level of immunity, from the virus.”
Another unknown is how protective a past COVID-19 infection combined with a first vaccine dose will be against new, highly transmissible variants. So, until further studies have been carried out, two doses remain essential.







