COVID-19 vaccines are now approved in some countries. What will it take to approve them for the rest of the world?
With the World Health Organization inviting COVID-19 vaccine manufacturers to submit their candidates for evaluation, we examine the process from submission to Emergency Use Listing.
- 2 February 2021
- 6 min read
- by Tetsekela Anyiam-Osigwe
A year after COVID-19 was declared a Public Health Emergency of International Concern (PHEIC), several vaccine candidates have now been developed and have already been made available to tens of millions of people, providing the world with a viable exit plan from the acute phase of the pandemic.
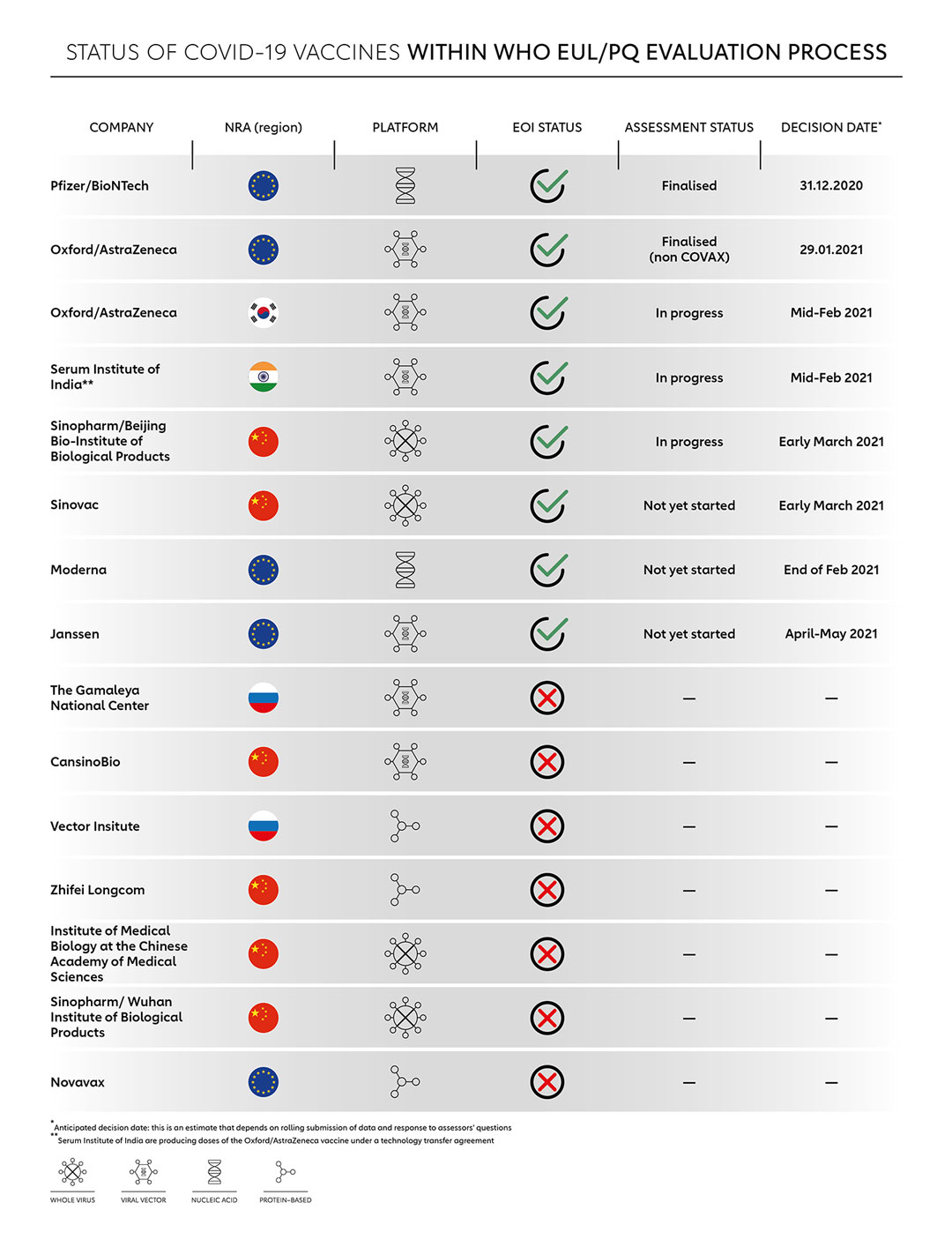
Globally, scientists and vaccine manufacturers have performed the seemingly impossible task of creating COVID-19 vaccines in less than a year through global collaboration, innovative technology and knowledge-sharing. Currently, over 60 COVID-19 vaccines are in clinical development, with at least 10 vaccines already approved by National Regulatory Authorities (NRAs) as well as regional authorities like the European Medicines Agency. Here’s what has to happen for these vaccines to be approved for worldwide use:
What does it take to get WHO approval?
Before a vaccine can be rolled out by any UN agency or vaccine-procuring international organisation, such as Gavi and the Global Fund, it needs to get WHO approval. This means going through a standard 'prequalification' procedure that often lasts several months in order to ensure that the requirements for safety and efficacy of immunisation programmes are adequately met. Once prequalification is completed, countries – especially those without any national regulatory authorities – can rely on this assessment to approve the deployment of the vaccine.
Have you read?
To qualify for WHO prequalification, or PQ, a vaccine must meet four criteria. First, it must be considered a priority for countries which are already part of vaccination programmes supported by UN agencies, like UNICEF. Second, it must be suitable for the public sector immunisation programmes in the countries where it is intended to be used. This – the programmatic suitability of the vaccine – often involves an assessment of thermostability/storage, dose volume, handling characteristics, labelling and packaging.
Third, the National Regulatory Authority (NRA) responsible for oversight of the vaccine must have full and adequate capacity, including for licensing, regulatory inspections, surveillance, laboratory access, as well as the authorisation and evaluation of clinical trials. In other words, WHO must have deemed the NRA to be ‘functional'. Lastly, the relevant NRA must have granted either a marketing authorisation or an emergency use authorisation for the vaccine. So far, no COVID-19 vaccine has been prequalified by the WHO.
The WHO EUL versus the WHO PQ
The Emergency Use Listing (EUL) procedure is not an alternative to WHO PQ, it is an entirely different process. The EUL is specifically tailored to public health emergency situations, like a pandemic, where data is likely to be limited and candidate vaccines cannot feasibly meet prequalification conditions. Whereas WHO PQ is targeted at the long-term introduction and use of vaccines, the circumstances characterising emergency situations – especially high morbidity and mortality rates – need a quicker pathway to get unlicensed vaccines time-limited approval for public delivery and use. The primary objective of the EUL is therefore to ensure that safe, effective and good quality vaccines are made available as quickly as possible to address the public health emergency. As part of the EUL, the WHO nonetheless expects the vaccine to be submitted for WHO PQ following full completion of vaccine development.
Since COVID-19 has been declared a global pandemic with no existing vaccines, any vaccine against COVID-19 developed in compliance with current Good Manufacturing Practices would be eligible for an EUL.
What happens after a vaccine is submitted to the WHO?
After a vaccine manufacturer submits an Expression of Interest for vaccine evaluation to WHO, it goes through these key stages before it can be granted an EUL:
Pre-submission meeting
Vaccine makers will be invited by WHO to a pre-submission meeting, during which there will be discussions over the assessment procedure to be used (EUL or PQ), the assessment pathway, the date of submission of the dossier, the submission package to be supplied and the availability of any supplemental data to support the application. For the manufacturer, these discussions are meant to help in making a final decision on submission. For WHO, it is an opportunity to coordinate resources to review the vaccine manufacturer’s dossier within the shortest possible amount of time.
Review and decision
Experts from individual national authorities are invited to participate in the EUL review as part of the ad-hoc Advisory Committee for Emergency Use and Listing (ACEUL). As the submitted COVID-19 vaccine would have already been assessed by a relevant NRA, the ACEUL will not be duplicating any of this work, which significantly shortens the review timeline. Instead, it will conduct review activities in areas where experts find it necessary to provide a level of assurance for vaccine safety, quality and efficacy. For example, critical programmatic aspects, such as dosage, autodisable syringes and storage temperature, are not assessed by the NRA of the manufacturing country because it falls outside their scope. The ACEUL will review such programmatic aspects, as well as data indicating vaccine safety and efficacy, as well as compliance with Good Manufacturing Practices.
Following the review, the ACEUL will issue a recommendation to WHO on whether the vaccine should be listed, and if so, under what conditions for use, in a limited time period. WHO will then use the recommendation of the ACEUL as a basis for its decision to grant the COVID-19 vaccine an EUL, which is valid for a maximum of 12 months.
Towards WHO regulatory approvals
By the fourth quarter of 2020, some COVID-19 vaccine manufacturers with vaccines in phase 2/3 clinical trials were preparing to seek approval from their National Regulatory Authorities. In October 2020, WHO invited these manufacturers to submit an expression of interest for either a WHO Emergency Use Listing (EUL) or Prequalification (PQ). Almost a month later, WHO followed this up by outlining its evaluation criteria for COVID-19 vaccines, which determines what pathway - either PQ or EUL - each vaccine would follow.
Being listed
In December 2020, the Pfizer/BioNTech vaccine against COVID-19 became the first vaccine to be granted an EUL from the WHO. This opened a pathway for countries who are yet to approve the vaccine to expedite their own national regulatory approval processes for it and for Gavi, alongside its Alliance partners, to procure and distribute the vaccine to participating countries through the COVAX Facility.
What happens post-listing?
After a product has been listed, WHO will continuously monitor field reports on safety surveillance, efficacy, and quality complaints about the COVID-19 vaccine – all of which may impact the validity of its EUL status. The COVID-19 vaccine manufacturer will also continue generating additional clinical data from vaccine trials and deployment to enable full licensure and WHO prequalification of its vaccine. This prequalification process usually takes a minimum of three months – only then can it be guaranteed that the vaccines, proven to be safe and effective, will be used in the long-term, with no regulatory barriers for them to be deployed in countries across the world.
More from Tetsekela Anyiam-Osigwe
Recommended for you