How a rapid-response bird flu vaccine trial is shaping global preparedness
As researchers in the UK launch a pivotal Phase 3 trial of Moderna’s mRNA based vaccine candidate against H5N1 bird flu, volunteer Clare Howard reflects on why she joined a CEPI supported effort to prepare the world for a rapid response to future pandemic flu outbreaks.
- 27 April 2026
- 4 min read
- by CEPI

At a health hub clinic in the southern English port city of Southampton, Clare Howard waits with anticipation as the nurse prepares her trial vaccination. She’s a little nervous, but is also buoyed by a sense of purpose. “It’s nice to think I’m doing something good,” she says. “Something that will benefit society.”
The experimental vaccine Clare is being dosed with is designed to protect against bird flu – one of the most serious and potentially fast-spreading known viral threats and one that is already circulating widely in wild birds, poultry flocks, cattle and other animals in many parts of the world.
Developed by the U.S. biotech company Moderna, the vaccine uses the same mRNA technology that underpinned its highly effective COVID‑19 shot. This candidate, mRNA‑1018, targets the A(H5N1) strain of bird flu.

With up to $54.3 million in funding from CEPI to help advance the vaccine to licensure and enable global equitable access if a pandemic were to strike, the shot is now being tested in a Phase 3 trial in the UK and the United States to assess whether it can safely trigger strong immune responses against H5N1.
"We know that the A(H5N1) strain is evolving and spreading across animal species, and though it does not yet move easily between humans, we have to treat human-to-human transmission as a real possibility,” said Dr Rebecca Clark, the trial’s national co-ordinating investigator based at Layton Medical Centre, Blackpool. “This trial is our proactive attempt to shield against that possibility, and any future pandemic that could emerge from it.”
Have you read?
According to data from the U.S. Centers of Disease Control and Prevention, there have been 116 confirmed human cases of H5 bird flu across the world just since 2024, almost all of them linked to close contact with infected animals.
That’s also why Clare, a physiotherapist who has her own hobby flock of four chickens at home in her back garden, volunteered to take part in the vaccine trial – to help the world be better prepared if a future human bird flu outbreak were to start to spread.
She often has to bring her chickens inside and keep them cooped up for many weeks when authorities send out alerts about avian flu outbreaks in wild birds or poultry in her area.
Clare light-heartedly refers to these incidents as “mini flockdowns”, but they are reminiscent of the deadly serious and life-changing lockdowns people across the world endured while they waited for new vaccines against COVID-19 to be developed. “If we can be more prepared and ready to go with a bird flu vaccine, that would be so much better for everyone,” she says.
Clare’s dose marks the UK start of the world’s first pivotal trial of an mRNA vaccine designed specifically for a potential influenza pandemic. Around 4,000 adults will take part across the UK and the United States, with most volunteers recruited through community clinics rather than traditional hospital sites.
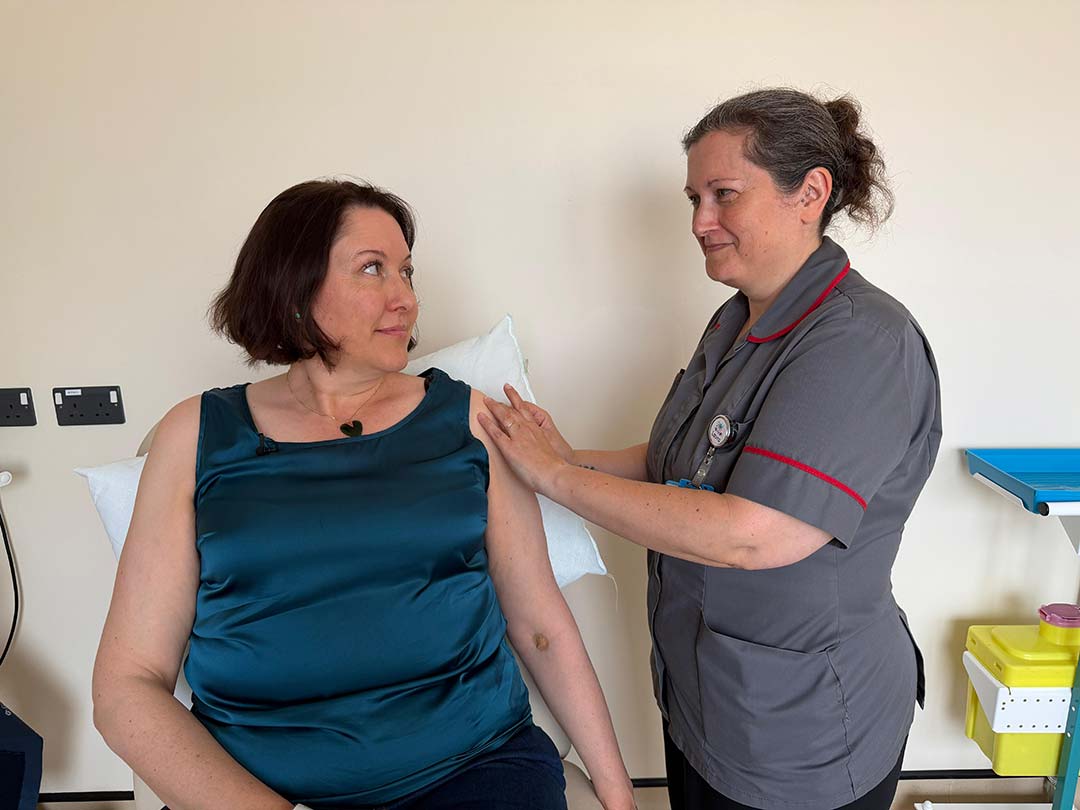
Dr Clark says the move to community trial sites is part of deliberate shift to bring vaccine research closer to where people live – an approach that proved positive during COVID‑19.
A central aim of CEPI’s investment in this bird flu vaccine development is equitable access. As part of the partnership, Moderna has pledged that 20 percent of its H5 pandemic vaccine manufacturing capacity will be allocated for timely supply to low- and middle‑income countries at affordable price levels if the vaccine is licensed.
“If successful, these efforts could transform our ability to respond swiftly and equitably to one of the world’s most enduring threats,” says CEPI’s CEO Dr Richard Hatchett.
As Clare Howard notes, being prepared for anything ahead of time is always better than having to play catch up with a deadly virus in the midst of a global pandemic.
“Novel viruses are just so dangerous,” she says. “With COVID, we didn’t have the time to prepare, so it’s great to be able to do something now that will contribute to protecting society.”