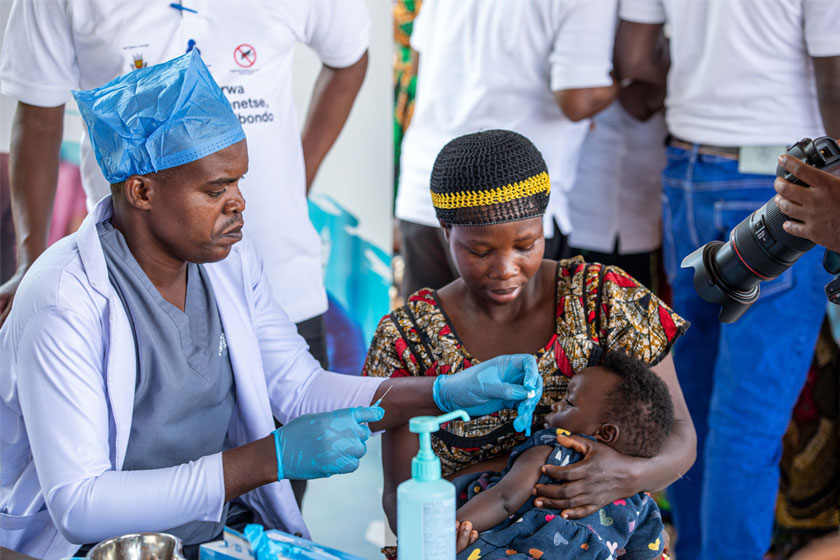
From rabies to hepatitis: Gavi to start rolling out new vaccines to lower-income countries
The pandemic meant several new Gavi vaccine programmes in lower-income countries had to be paused. Following a Board decision this week, these programmes are back on.
- 28 June 2023
- 7 min read
- by Gavi Staff

The COVID-19 pandemic halted the roll-out of many essential vaccines, and catch-up campaigns are in place to continue these pre-pandemic efforts. Gavi has a Vaccine Investment Strategy (VIS), in which new or underused vaccines are made available to lower-income countries across the world. This strategy is revised every five years and the last VIS process was completed in 2018.
The impact of these vaccine rollouts can be life-changing often, without support, countries are unable to provide these vaccines to their populations, and Gavi funding has allowed, for instance, the set-up of HPV vaccine programmes aimed at preventing cervical cancer in women and the stockpiling of cholera vaccines for emergencies.
In September 2020, the Gavi Board paused the availability of funding for vaccines that had been approved for inclusion. This included rabies vaccine hepatitis B birth dose diphtheria, tetanus and pertussis (DTP)-containing boosters.
Now these vaccine roll-outs are to begin again, since countries are regaining the capacity and resources to implement vaccine delivery programmes other than COVID-19 vaccines. In addition multivalent meningococcal vaccine and respiratory syncytial virus (RSV) vaccine were also both approved in 2018, but are only reaching the stage where vaccines are available and approved for rollout.
Here's why these vaccines are important and the impact they will have across the world.
Hepatitis B birth dose
Potential impact: Could prevent up to 1.5 million cases and up to 1.2 million perinatal infection-related deaths over 15 years
Hepatitis B virus is 100 times more infectious than HIV and kills 884,000 people a year, thanks to a highly effective virus that can cause chronic inflammation in the liver. Babies and infants are most at risk, and nine out of ten infants who are infected will go on to develop chronic hepatitis B. A quarter will develop severe liver disease.
Vaccination is critical, because while the virus can be transmitted in body fluids, such as blood or semen, many people become infected in the womb when their mother is carrying the virus. Children can be silent carriers of the infection that doesn't become symptomatic until their 40s or 50s , when they discover they have liver failure (as the inflammation causes cirrhosis) or liver cancer.
The current hepatitis B vaccine is a protein-based subunit vaccine, containing the HBV surface antigen (HBsAg) protein that sits on the surface of the virus. The World Health Organization (WHO) recommends that all babies receive the vaccine as soon as possible after birth, preferably within 24 hours, followed by a further two or three doses at least four weeks apart. Ensuring a reliable vaccination programme is important as children need three doses to be protected, but once they are, it lasts at least 20 years and is probably lifelong.
Rabies
Potential impact: Could save up to 267,000 lives
The rabies virus is spread through the bite of infected animals, most often dogs (99% of rabies infections happen this way), but bats, raccoons and foxes can transmit it to people too. Generally people become infected through getting bitten, but in rare cases, scratches or broken skin can get infected if the person comes into contact with the saliva or other bodily fluids of an infected animal.
The disease kills tens of thousands of people every year. While rabies is present worldwide, 95% of deaths are in Africa and Asia, and most are children younger than 15 years as they are most likely to be outside and coming into contact with rabid dogs.
Once someone is symptomatic the disease is 100% fatal, meaning that vaccination in high-risk areas is critical, especially as access to treatment with immunoglobulins is unlikely to be available.
The vaccine can be given as pre-exposure prophylaxis (i.e. before a person is bitten) but it can also be given immediately after someone has been bitten, and it is extremely effective – almost 100%. For people who are at high risk, booster doses are recommended, whether or not they have ever been bitten. Gavi will support the post-exposure prophylaxis strategy: a one-week, three-dose regimen.
The decision to roll out the vaccine is supported by the strong global momentum of the Zero by 30 campaign, led by WHO, the Food and Agriculture Organization (FAO), World Organisation for Animal Health (WOAH, formerly OIE) and the Global Alliance for Rabies Control (GARC) with the goal of eliminating dog-mediated human rabies by 2030.
Multivalent meningococcal
Potential impact: Could save up to 106,000 lives, and prevent up to one million cases
Meningococcal disease causes hundreds of thousands of cases every year, with around 89% of deaths in Gavi countries. These mainly take place in sub-Saharan Africa's "meningitis belt" of 26 countries.
Bacterial meningitis can be caused by multiple serogroups of the bacterium Neisseria meningitidis (and can also be caused by pneumococcal and streptococcal viruses) and can kill often and with speed – about one in ten people who get a severe case die, sometimes within a few hours of developing symptoms.
The inflammation that develops around the brain means that around one in five may develop severe complications, such as hearing or vision loss, epilepsy, problems with memory, concentration, movement or balance, loss of limbs, or bone, joint and kidney problems.
Babies with meningitis might be irritable, or extremely sleepy. They also often have a tell-tale rash that starts as small, red pinpricks turning into red or purple blotches that don't fade in response to pressure.
Have you read?
While meningococcal disease has historically been dominated by the A subgroup and prevented by the meningococcal A conjugate vaccine, other serogroups C, W, and X have recently become more prevalent, making multivalent vaccines much more important.
Four tetravalent ACWY conjugate vaccines exist on the market at high cost additional tetravalent vaccines are in the pipeline and could be available at lower cost in the future. A prequalified pentavalent conjugate vaccine is expected to become available at a lower price than current tetravalent vaccines.
Diphtheria, tetanus and pertussis (DTP)-containing boosters
Potential impact: Booster doses could save 106,000 lives, 82% of which would be in children under 5 years old
Together, these three pathogens kill around 110,000 people a year – 65% of deaths due to pertussis, 34% due to tetanus, and 1% due to diphtheria. The greatest burden is in Asia and sub-Saharan Africa and most deaths occur in children aged one to four years.
In 1948, a combination vaccine against diphtheria, tetanus pertussis began to be rolled out. It's recommended to be given to babies at two, four, six and 18 months of age and then again at four to six years old, followed by a booster dose every ten years. Gavi has rolled these vaccines out in a pentavalent vaccine that also includes hepatitis B and Hib immunity from primary pentavalent series wanes to non-protective levels after around three to five years against the three diseases (assuming three doses of the primary series).
In 2018, as an extension to Gavi's existing pentavalent vaccine programme, the Board decided to support routine boosters in ages 12 to 23 months (diphtheria, tetanus and whole cell-pertussis [DTwP] or pentavalent), four to seven years (tetanus and diphtheria [Td] – containing) and nine to 15 years (Td-containing), according to WHO's recommendation.
Respiratory syncytial virus
Potential impact: a maternal vaccine could save up to 206,000 lives and prevent up to 10.4 million cases
Lower respiratory tract infections (LRTI) kill more infants than malaria or diarrhoea combined and respiratory syncytial virus (RSV) is one of the major causes, with the majority of deaths occurring in lower-income countries among previously healthy children. RSV infects millions each year, but it's the immune systems of babies and elderly people that are most vulnerable.
The virus is estimated to cause 33 million cases annually and 3.2 million related hospitalisations in children under five years old, resulting in 77,000 deaths per year. Ninety percent of children affected are younger than five years, with mortality highest in babies under six months.
Until now there hasn't been an approved vaccine, though the US Federal Drug Administration (FDA) has just approved the world's first vaccine for use in people over 60, which can reduce the risk of severe RSV-related lower respiratory disease by 94% in the RSV season following vaccination.
Prophylactic monoclonal antibodies are already approved to protect at-risk babies, but these are not available in low-income countries, where babies will need to rely on a vaccine. Palivizumab is widely approved across Europe, US, Canada, Asia, and Latin America, but recommendations for its use vary from country to country. Given its high price ($1,000-$2,000 per dose) and the need to administer it monthly during the RSV season, its use is restricted to patients with high-risk pathologies, mainly in high-income countries (HIC)
A maternal RSV vaccine is currently in Phase III trials and will be considered for approval. An interim analysis indicated over 80% efficacy against medically-attended severe RSV-associated lower-respiratory tract infection during the first three months of life.