Gavi’s impact
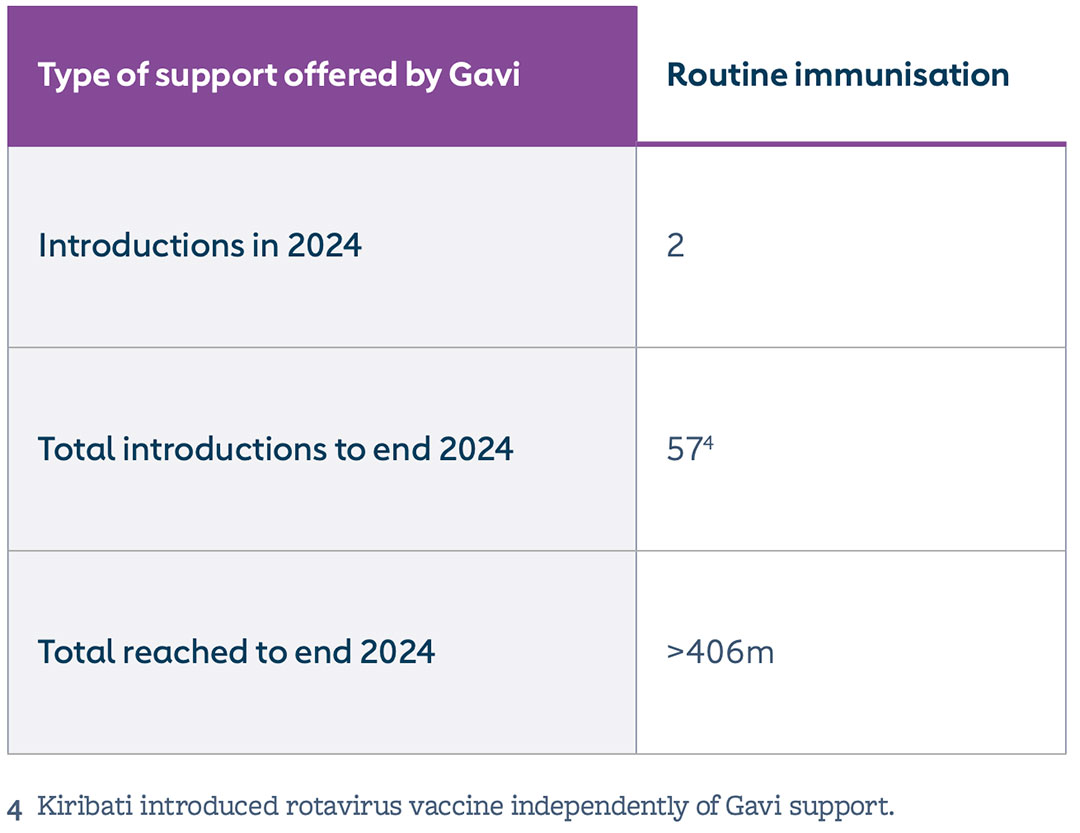
By end 2024, more than 406 million children had been immunised with rotavirus vaccine with Gavi support. Rotavirus vaccine coverage across the 57 Gavi-supported countries increased by 2 percentage points in 2024, reaching 71%. In 2024, Chad and Viet Nam introduced rotavirus vaccine into the routine immunisation programme, further closing the gap in rotavirus vaccine routine introductions in Gavi-eligible countries. Additionally, South Sudan’s and Cambodia’s requests for routine introduction of rotavirus vaccine were approved.
Somalia, South Sudan and Cambodia initiated planning with support from partners and will introduce rotavirus vaccine in 2025. In 2024, two manufacturer-driven supply disruptions occurred due to product discontinuations, requiring affected countries to transition to alternative products to avoid stock-outs.
The issue
Rotavirus disease is the main cause of infant deaths from diarrhoeal disease globally.
DISEASE BURDEN
Rotavirus is a virus that can cause inflammation of the stomach and intestines. Symptoms include severe watery diarrhoea, often with vomiting, fever, and abdominal pain. Infants and young children are most likely to get rotavirus disease.
In serious cases, children risk dying from dehydration. Around 443,832 children under five years die from diarrhoeal disease each year; it’s the third leading cause of death in children aged under five years. Approximately half the global total of rotavirus deaths occur in Africa and South-East Asia.
PREVENTION
Unlike other types of diarrhoea, improving hygiene does not prevent rotavirus infection. Also, antibiotics and other drugs cannot cure rotavirus. Therefore, vaccination is the best way to prevent rotavirus illness and death. WHO recommends that all countries introduce rotavirus vaccines in their national programmes; and emphasises the need to integrate rotavirus vaccine with other interventions.
Gavi’s response
Gavi offers support for lower-income countries to introduce rotavirus vaccines. Gavi funds the cost to procure the rotavirus vaccine itself, as well as a one-time grant to cover introduction costs.
In 2009, the WHO Strategic Advisory Group of Experts on Immunization (SAGE) recommended the universal rotavirus vaccination of infants. Following this decision, Gavi started to offer this type of support worldwide. This allowed for countries in Africa and Asia, where most rotavirus deaths occur, to introduce the vaccine. The WHO position paper on rotavirus vaccines was updated in 2021 and continued to recommend the inclusion of rotavirus vaccination in all national immunization programmes.
FROM GAVI'S 2024 ANNUAL PROGRESS REPORT:
From other sites
- WHO | Immunization surveillance, assessment and monitoring
- WHO | Immunization, Vaccine and Biologicals
- WHO | Initiative for Vaccine Research: Diarrhoeal Diseases
- WHO | New and Under-utilized Vaccines Implementation
- WHO | Vaccine Safety
Latest articles about rotavirus
View moreNew vaccines make big impact in rural Uzbekistan
Pneumococcal and rotavirus vaccines are not only shielding children from disease, they’re building confidence in vaccination in the country’s remotest corners.
Q&A: ‘India must ready tools to prevent virus outbreaks’
From rotaviruses to flu, vaccines are key to protecting the vulnerable and preventing future pandemics, says India’s Gagandeep Kang.
Mozambique revs up rotavirus vaccination in rural Zambezia province
Rotavirus vaccine coverage took a hit during the pandemic in populous Zambezia province, Mozambique. Now authorities are racing to catch up.