Hib Initiative: a GAVI success story
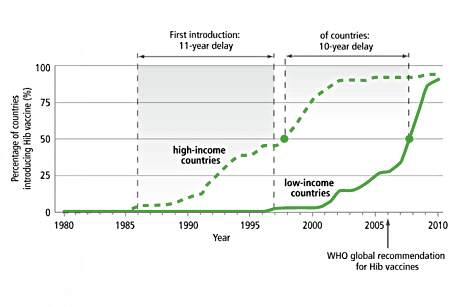
Hib vaccine introduction in high- and low-income countries. Source: WHO, Vaccine introduction database 2010 Jan 22 [cited 2011 May].
By the late nineties, half of all high-income countries were routinely using vaccines against Haemophilus influenzae type b (Hib) infection. But the developing world was lagging far behind.
High costs meant low-income countries had no access to the Hib vaccine with the sole exception of The Gambia which, thanks to a donation from the manufacturer, had added the vaccine to its national immunisation programme in 1997 -- 11 years after Canada became the first high-income country to introduce routine Hib vaccination.
When GAVI was set-up in 2000, support for Hib vaccine was immediately offered through its new and underused vaccine support (NVS). However, uptake was slow due in part to a lack of understanding of the full burden of the problem.
Barriers to vaccine introduction were thought to be limited awareness and communication about the disease, uncertainty about Hib disease burden, and concerns about the cost of the vaccines.1
Dedicated campaign needed
It took a dedicated campaign from GAVI to bring positive and dramatic change.
In 2005, the GAVI Board allocated a four-year US$ 37 million grant to set up the Hib Initiative to catalyse the vaccine's uptake.
By pooling the knowledge and expertise of the Johns Hopkins Bloomberg School of Public Health, the London School of Hygiene & Tropical Medicine, and the Centers for Disease Control and Prevention (CDC), the Hib Initiative used a combination of collecting and disseminating existing data, research and advocacy to help countries build a case for adopting the Hib vaccine.
Widespread success
Coupled with the WHO recommendation in 2006 that Hib vaccines should be included in every national immunisation programme, the Initiative was a rapid and widespread success.
In 2008 - 10 years after high-income countries crossed the 50 percent line - the vaccine had been introduced in half of all low-income countries. By 2009, 62 countries, nearly all low-income countries, had introduced Hib vaccines with GAVI support, preventing an estimated 430,000 future deaths.
GAVI's subsidisation of the combination pentavalent vaccine has also encouraged uptake by allowing low-income countries to introduce the Hib vaccine at the same time as protecting their children against four other diseases: diphtheria-tetanus-pertussis (DTP3) and hepatitis B (hepB).
Hib meningitis eradicated in many sub-Saharan countries
A 2008 study estimated that, within four years of GAVI support helping Uganda to introduce Hib vaccine into the national vaccination programme (June 2002), the incidence of Hib meningitis declined by 85 percent, and by the following year, the number of cases fell to nearly zero.2
The impact of Hib vaccine inroduction has also been demonstrated in other GAVI eligible countries including Kenya3, Malawi4 and Senegal5.
1 Hib Initiative. Supporting country decision making regarding hib vaccine use: strategic plan. Baltimore: Hib Initiative. 2006 [cited 2011 May]
2 Lee Eh, Lewis RF, Makumbi I, Kekitiinwa A, Ediamu TD, Bazibu M, et al. Haemophilus influenzae type b conjugate vaccine is highly effective in the Ugandan routine immunization program: a case-control study. Trop Med Int Health. 2008 Apr; 13(4):495-502
3 Cowgill KD, Ndiritu M, Nyiro J, Slack MP, Chiphatsi S, Ismail A, et al. Effectiveness of Haemophilus influenzae type b conjugate vaccine introduction into routine childhood immunization in Kenya. JAMA. 2006 Aug 9; 296(6):671-8.
4 Daza P, Banda R, Misoya K, Katsulukuta A, Gessner BD, Katsande R, et al. The impact of routine infant immunization with Haemophilus influenzae type b conjugate vaccine in Malawi, a country with high human immunodeficiency virus prevalence. Vaccine. 2006 Sep 11; 24:6232-39.
5 Cisse MF, Breugelmans JG, Ba M, Diop MB, Faye PC, Mhlanga B, et al. The elimination of Haemophilus influenzae type b meningitis following conjugate vaccine introduction in Senegal. Pediatric Infect Dis J. 2010 June; 29(6). Epub 2009 Dec 29.
