Gavi’s market shaping efforts aim to make life-saving vaccines and other immunisation products more accessible and affordable for lower-income countries.
Healthy markets for vaccines and other immunisation products allow manufacturers to plan production based on known demand, donors to maximise their investments and, most importantly, lower- and middle-income countries to buy suitable products at prices they can afford.
Market failures
Market forces do not always serve low-income countries well. Historically, uncertain funding and demand for vaccines led to manufacturers not having the incentive to invest in new products at affordable prices for developing countries.
As a result, there was often a long time lag between the time a vaccine was available in rich countries and when it was introduced in LMIC countries.
OUR SOLUTION
Gavi was created to address these market failures. Gavi’s fourth strategic goal to ensure sustainable, healthy market dynamics for immunisation-related product and to incentivise innovation seeks to further Gavi’s mission to “leave no-one behind with immunisation.” Through our Market Shaping strategy, we make proactive efforts to improve the health of markets for vaccines and other immunisation products.
PRIORITIES
The market shaping strategy has three priorities:
- Fostering a sustainable competitive future supplier base;
- Healthy demand driven by a supportive environment; and
- Establishing an enabling environment for transformational innovation.
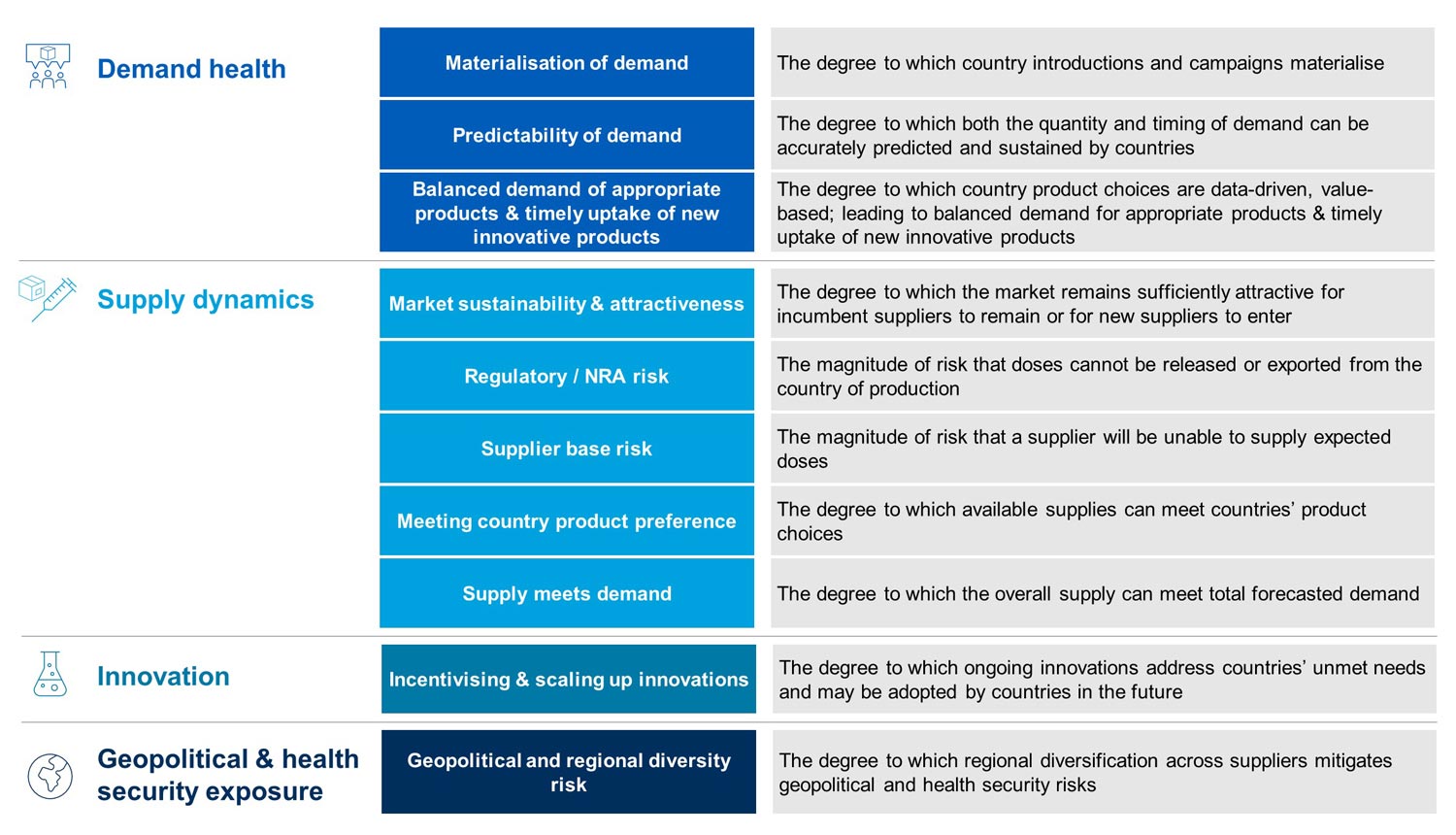
The healthy markets framework
In 2015, the Gavi Secretariat, UNICEF and the Gates Foundation jointly developed a healthy markets framework (HMF).
The framework helps us to develop common strategies for vaccine markets across the Vaccine Alliance through the development of a clear and consistent definition of market health. Its main aims include: establishing a common way of thinking about market health, communicating Alliance assessments of individual markets, and improving trade-offs between how different market elements are analysed.
For the 2021–2025 period, the Healthy Markets Framework assesses overall market health by taking demand health directly into account, in addition to doing so through supply dynamics and innovation perspectives. The new iteration of the HMF is also designed to reflect a greater emphasis on long-term market views, in alignment with the Gavi’s overall objectives for this period. Additionally, as part of Gavi’s regional manufacturing strategy, the HMF was updated in 2024 to include a new dimension on the degree to which regional diversification across suppliers mitigates geopolitical and health security risks.

Regional manufacturing strategy
Our regional manufacturing strategy (RMS) was developed to respond to the challenge surrounding equitable access to vaccines during the COVID-19 pandemic, and to the African Union’s call to action on building regional manufacturing capacity in Africa. This includes the African Vaccine Manufacturing Accelerator (AVMA), a financing mechanism established to accelerate the expansion of commercially viable vaccine manufacturing in Africa.

Vaccine demand forecasting
Long-term strategic demand scenarios offer visibility into future market needs to Gavi stakeholders.

Market Shaping roadmaps
Market Shaping roadmaps help guide Gavi’s market shaping efforts by analysing the dynamics of each vaccine or other immunisation product market.
Product information for vaccines and cold chain equipment
Detailed product profiles and the Cold Chain Equipment Technology Guide help countries decide which vaccine presentations or cold chain equipment are the best “fit” for their immunisation programmes.

The Vaccine Innovation Prioritisation Strategy
VIPS represents an unprecedented three-year collaboration between the Gavi Secretariat, World Health Organization (WHO), Gates Foundation (GF), United Nations Children’s Fund (UNICEF) and PATH – known as the VIPS Alliance – to develop a single integrated framework to evaluate and prioritise vaccine product innovations and to drive these innovations forward.