Contents
Benefits of the hexavalent programme
Documents
Gavi support guidelines
Contact
Interested Gavi-eligible countries should contact their Senior Country Manager. For general inquires, please email info@gavi.org with the words "Hexavalent vaccine" in the Subject line of your email.
Share
Eligible countries may apply for hexavalent vaccine
Programme update
Starting 1 December 2023, countries eligible for Gavi support can apply to switch to a whole-cell pertussis (wP) hexavalent vaccine (hexavalent) – a six-in-one vaccine that combines the pentavalent vaccine (diphtheria, tetanus, whole-cell pertussis [DTwP], hepatitis B and Haemophilus influenzae type b) with inactivated polio vaccine (IPV).
Hexavalent vaccine is expected to help countries deliver protection against all six diseases more efficiently and cost-effectively – as well as reduce programmatic challenges due to multiple injections, among other anticipated programmatic benefits.
In Gavi’s 2026–2030 strategic period (Gavi 6.0), hexavalent vaccine is categorised under the “Guaranteed budget” component. Countries planning to switch to hexavalent vaccine in Gavi 6.0 are required to include it in their forecast as well as holistic application. If a country chooses not to introduce or maintain a vaccine in its guaranteed bucket under Gavi 6.0, the associated guaranteed funding is withdrawn and reallocated, as those funds cannot be retained or repurposed unless the country self‑finances the programme.
Countries can choose to switch to hexavalent or continue using pentavalent and IPV. Vaccine Alliance partners can assist countries to assess the financial, logistical and programmatic implications of a switch to hexavalent to determine which option is most suitable for their context. Countries will need to continue using bivalent oral polio vaccine (bOPV) in their routine immunisation programmes until global cessation. Hexavalent contains IPV but not bOPV, and both are needed.
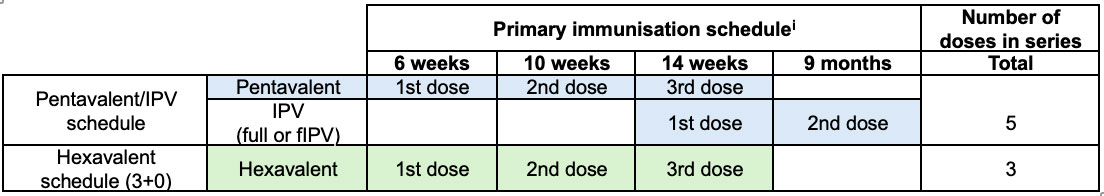
Hexavalent schedule
Starting from 6 weeks of age (minimum), the World Health Organization (WHO) recommends a hexavalent vaccine schedule of 3 doses with a minimum interval of 4 weeks between doses. Illustrative schedules are shown below.
Benefits of the hexavalent programme
Programmatic efficiency
- Reduces number of injections: By combining pentavalent and IPV in the same vaccine, it reduces the number of injections and eases the burden on children, caregivers and health care workers – particularly in the context of vaccine hesitancy. This may also make it easier to reach zero-dose and under-immunised children.
- Reduces costs: Fewer doses administered lead to cost savings on syringes, safety boxes, cold-chain, in-country transportation, associated energy consumption and labour.
Support for polio eradication
- Facilitates IPV coverage: Combining IPV with pentavalent increases opportunities for under-immunised children to receive sufficient IPV doses.
- Facilitates integration of IPV: It is anticipated that IPV will remain in routine immunisation long after certification of polio eradication. Providing IPV as part of a combination vaccine with other core antigens ensures streamlined integration into national immunisation programmes.
Gavi vaccine and programme funding for hexavalent
- From 2024, countries eligible for Gavi support will have gradual access to hexavalent vaccine at the price of US$ 2.85 per dose. The cost per dose is expected to decline in the near future, approaching price parity with standalone IPV and pentavalent vaccines.
- Gavi-eligible countries that decide to switch to hexavalent must co-finance this vaccine based on the regular Gavi co-financing policy; and they will continue to benefit from the IPV co-financing exemptions as described in the table below.
Operational support is available under the consolidated cash grant.
| Initial self-financing (ISF) | Preparatory transition (PT) | Accelerated transition (AT) | Fully self financing | ||
|---|---|---|---|---|---|
| Lower middle-income country | Upper middle-income country | ||||
| 3xpenta | 3 doses x US$ 0.20/dose | Co-financed percentage of [3 doses x penta] | 50% support in 2027 Full self-financing from 2028 | Full self-financing from 2026 | |
| 2xIPV | US$ 0.00 | US$ 0.00 → US$ 0.60/childii | |||
| 3xhexa | 3 doses x US$ 0.20/dose | Co-financed percentage of [4 doses x hexa at US$ 2.85/dose minus value of IPV funded by Gavi] | |||
Special considerations for switching to hexavalent
- The feasibility and financial implications (including savings) of switching to hexavalent: Countries may pay more in vaccine costs for the hexavalent 3-dose series than for the current 3-dose pentavalent + 2-dose IPV, but they would benefit from programme cost savings in delivery (e.g. reduced injections, health care worker labour).
- 10-dose presentation: Initially, hexavalent will only be available to Gavi-eligible countries in a 10-dose presentation. Countries will need to consider the programmatic implications (e.g. cold chain, wastage) of using 10-dose vials if they are currently using a 1-dose presentation for their pentavalent product.
- Planning and implementation logistics: Countries will need to switch from pentavalent to hexavalent; phase out of standalone IPV and pentavalent.
- Introduction of a second dose of IPV (IPV2): WHO recommends that countries introduce IPV2; those that have not done so yet are encouraged to introduce hexavalent or standalone IPV2 as a matter of urgency.

