Gavi's impact
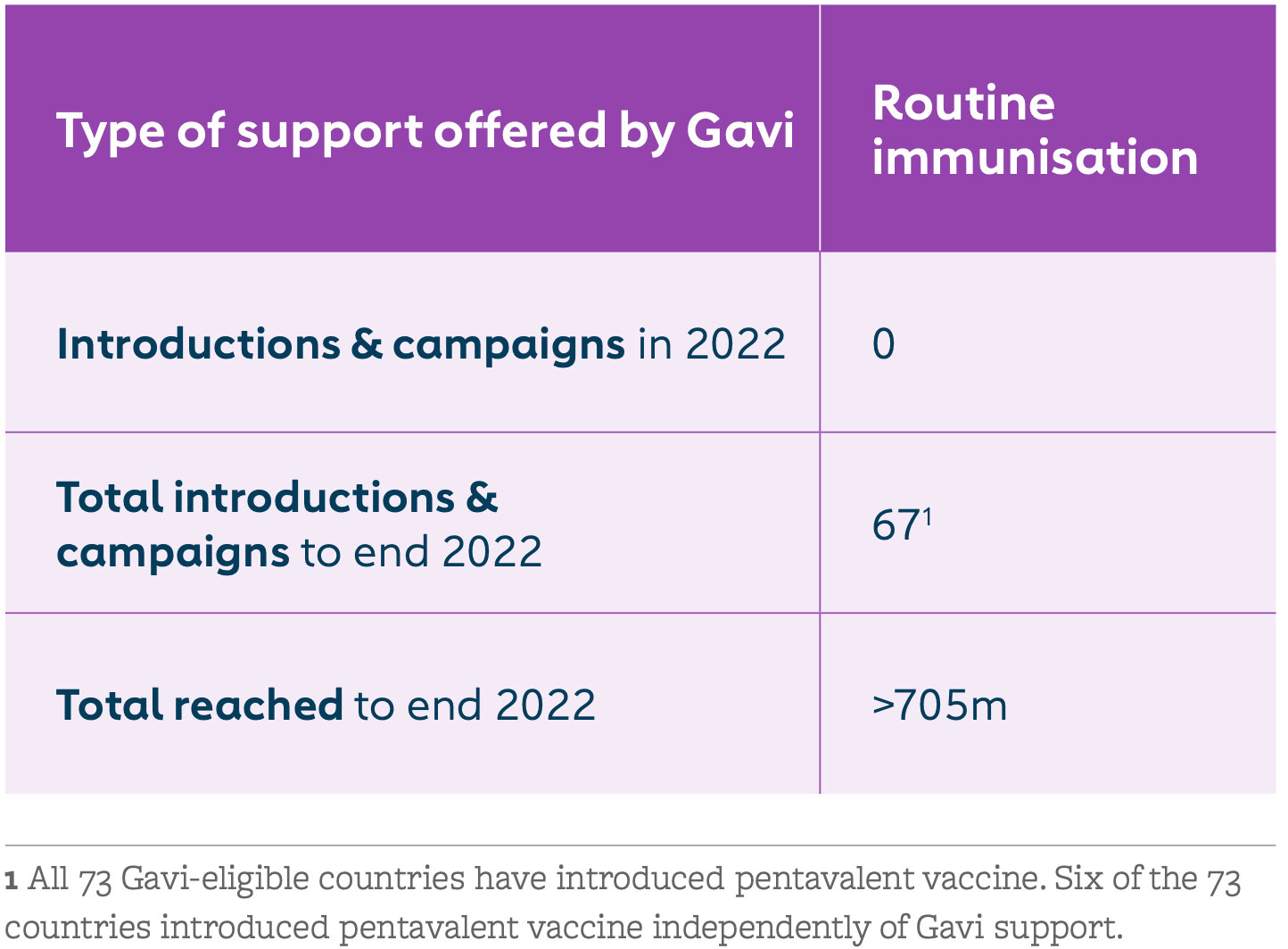
Pentavalent vaccine coverage in the 57 Gavi-supported countries increased from 0% in 2000 to 81% in 2022. While there were setbacks in coverage during the COVID-19 pandemic, coverage rates for the first and third doses of pentavalent vaccine are almost back to pre-pandemic levels. Efforts by Vaccine Alliance core and expanded partners to operationalise the zero-dose agenda have facilitated these improvements. By end 2022, more than 705 million children had been immunised with three doses of Gavi-funded pentavalent vaccine.
The issue
The pentavalent vaccine is vital to the survival of children aged under five. It is a five-in-one combination vaccine that protects these children from the following diseases:
DIPHTHERIA
A highly contagious bacterial infection that spreads from person to person, often through respiratory droplets, like from coughing or sneezing. Globally, incidence of diphtheria is on the rise. Death occurs in 5% to 10% of those infected, mainly in children aged under five.
TETANUS
A serious, life-threatening disease that affects the central nervous system and enters the body through wounds or cuts. Neonatal tetanus continues to be a major killer of newborns and can be prevented by providing the vaccine to women of childbearing age, either before or during pregnancy.
PERTUSSIS (WHOOPING COUGH)
A highly contagious and serious respiratory disease that spreads easily from infected persons and is fatal in 1 in 200 cases among infants.
HAEMOPHILUS INFLUENZAE TYPE B
A deadly bacterium that can cause meningitis, pneumonia and septicaemia – spread through sneezing and coughing. Hib is the third-leading vaccine-preventable cause of death in under-fives. In lower-income countries, where most Hib deaths occur, the disease leaves up to 35% of survivors with disabilities.
HEPATITIS B
A viral infection that affects the liver and is transmitted by body fluids or the blood of infected people. HepB is the leading cause of liver cancer and is 50 times more infectious than HIV. Transmission of the virus from mother to newborn infant is a major contribution to disease in regions such as South-East Asia and the Western Pacific, where infection is widespread. Most cases could be avoided through vaccination.
Gavi's response
Gavi began supporting pentavalent vaccine in 2001. The goal was to boost the low uptake of Hib and hepB vaccines by making them part of routine immunisation programmes in low-income countries. Since 2012, Gavi has supported Hib and hepB vaccines only as part of the pentavalent vaccine. With our support, the pentavalent vaccine is now available in the world’s 73 lowest-income countries.
Countries have seen remarkable benefits from this combination vaccine, including cost savings, improved programmatic efficiencies, increased coverage and equity, and reduced environmental impact – among other advantages.
The pentavalent vaccine success story reflects the strengths of Gavi’s public-private partnership model. UNICEF Supply Division has met demand for over 1 billion doses; WHO and UNICEF helped countries decide when and how to introduce the vaccine; and industry has increased global production capacity from 20 to 600 million doses. At the same time, innovation in formulation and packaging has helped to reduce the strain on immunisation cold chains.
Starting 1 December 2023, countries eligible for Gavi support can apply to switch to a whole-cell pertussis (wP) hexavalent vaccine – a six-in-one vaccine that combines the pentavalent antigens with inactivated polio vaccine (IPV) – or continue using pentavalent and IPV standalone vaccines. At this time, eligible countries can also apply for support to introduce any of the three WHO-recommended DTP-containing vaccine boosters in the national immunisation schedule (12–23 months, 4–7 years and 9–15 years).
FROM OTHER SITES
- Summary of WHO Position Papers - Recommended Routine Immunizations for Children
- WHO position paper on diphtheria vaccine
- WHO position paper on tetanus vaccine
- WHO position papers on pertussis vaccines
- Pentavalent vaccine (DTwP-HepB-Hib): Market & Supply Update
- MSF medical guidelines
- Protecting all against tetanus: guide to sustaining maternal and neonatal tetanus elimination (MNTE) and broadening tetanus protection for all populations
From Gavi's 2022 Annual Progress Report:
Related articles
View moreTogo has a cancer problem, but the government says vaccines will help
In 2022, nearly two thirds of the Togolese people diagnosed with cancer died. Vaccination can help change those figures, say Ministry of Health officials.
How combining vaccines saves money and lives
New vaccines are urgently needed, but fitting them into increasingly crowded immunisation schedules is challenging. Could combination vaccines provide a solution?
6 things to know about the 100-day cough
Whooping cough or pertussis – also called the 100-day cough – is on the rise in the UK and US. Here’s what you need to know.