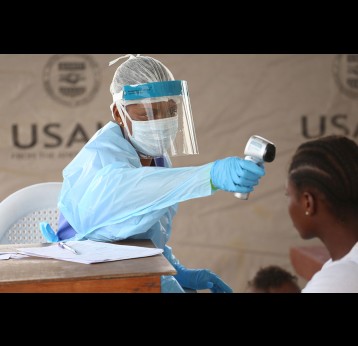
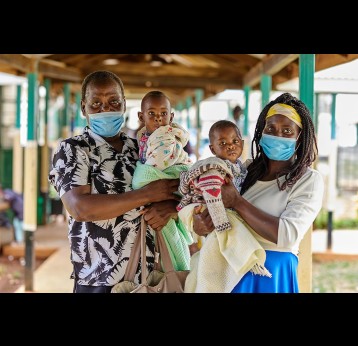
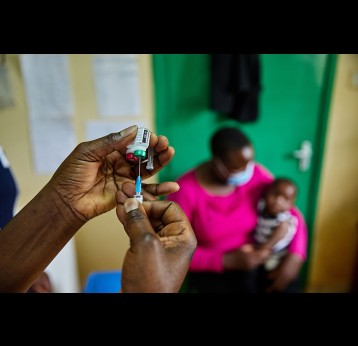
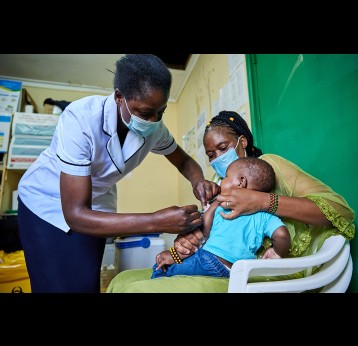
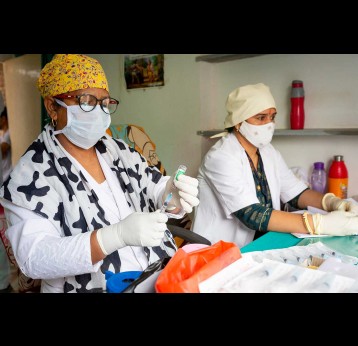
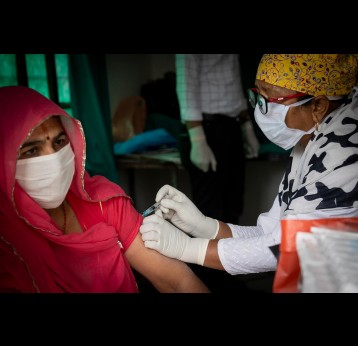
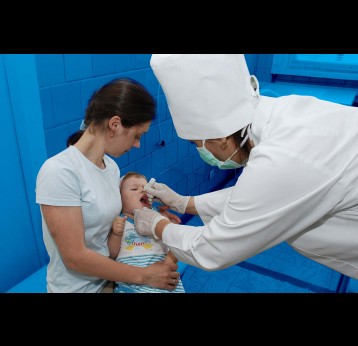
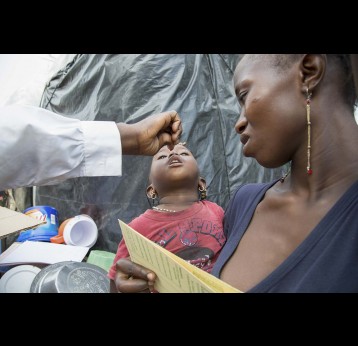
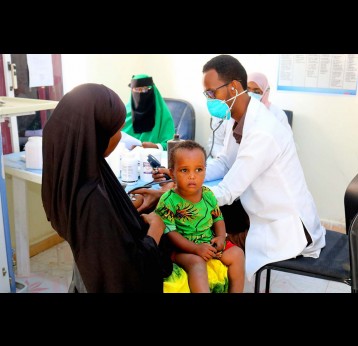
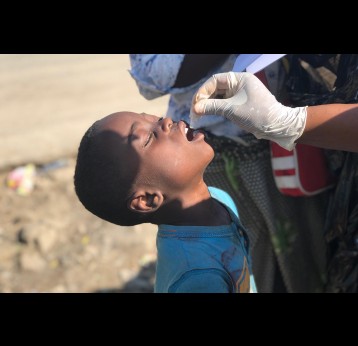
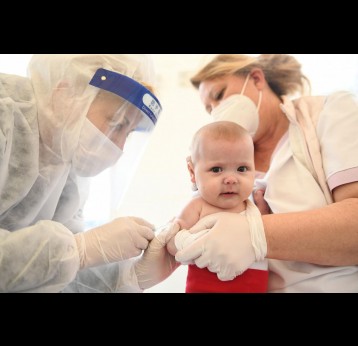
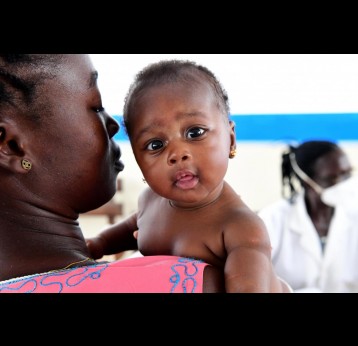
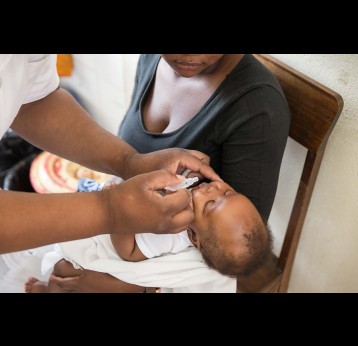
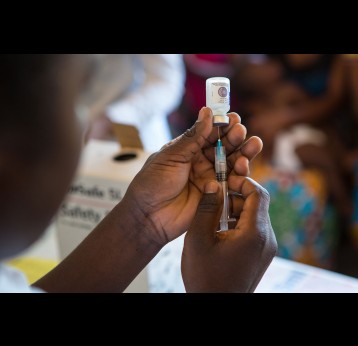
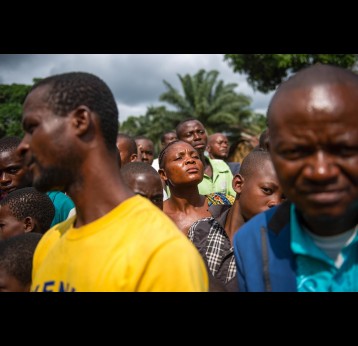The last mile of polio eradication and the vaccination challenges it brings
We are close to a polio-free world, yet cases of vaccine-derived disease are challenging the eradication effort.
- 28 September 2020
- 6 min read
- by Priya Joi
-
Republish this article
Disclaimer
If you would like to republish this article, please follow these steps: use the HTML below; do not edit the text; include the author’s byline; credit VaccinesWork as the original source; and include the page view counter script.

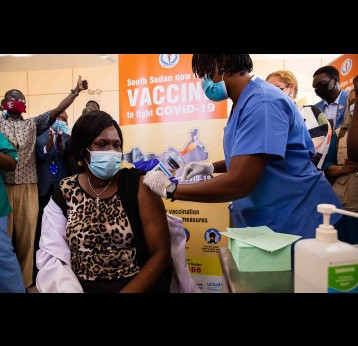
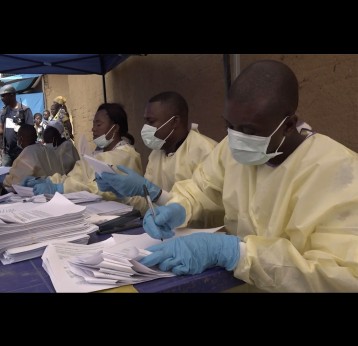
The effort to eradicate polio is close to the finish line. In 2019 there were 542 cases, down from 350,000 in 1988. Polio would become the second disease that we’ve managed to eradicate since smallpox in 1980. It is important to note that two of the three types of wild poliovirus (WPV) have already been eradicated globally and it is only type 1 poliovirus that continues to circulate in just two remaining countries, Afghanistan and Pakistan. An historic milestone was reached earlier this month when the World Health Organization (WHO) announced that wild poliovirus had been eradicated in Africa. Shortly after however came news of yet another outbreak in Sudan of circulating vaccine derived poliovirus (cVDPV). Sudan’s last confirmed case of WPV was in March 2009 but similar to other countries in Africa and in other regions of the world, is now faced with responding to a polio outbreak that is linked to the oral polio vaccine (OPV) that uses a live but weakened form of the virus.
1. How does OPV lead to cases of polio in some instances?
As the vaccine-virus is shed by vaccinated individuals, it can occasionally change its genetic make-up and spread in communities that are not fully vaccinated against polio, especially in areas where there is poor hygiene, poor sanitation or overcrowding. The vaccine-virus can continue to spread in an under-immunised population and recombine with other circulating vaccine viruses.
Over time, the attenuated vaccine virus can change to the point where it regains the ability to cause damage to the nervous system and paralysis, giving rise to a cVDPV. This is more likely to occur in areas where the vaccine-virus is allowed to continuously spread and is therefore associated with poor sanitation, hygiene or overcrowding and chronically low levels of population immunity to polio.
2. Why do countries continue to use OPV?
Although wild poliovirus circulation is now limited to Afghanistan and Pakistan, the risk of international spread remains, and is the basis for continued use of oral polio vaccines. Unlike IPV (inactivated poliovirus vaccine) – that has recently been introduced into routine immunisation programmes in Gavi-supported countries – oral vaccines are made from a live but weakened strain of the virus. OPV is capable of inducing mucosal immunity in the intestine, the primary site where poliovirus replicates. By doing so, the vaccine prevents shedding of the virus into the environment and can limit or stop person-to-person transmission. This is especially important in communities with poor water and sanitation infrastructure where people are more likely to be exposed to water-borne pathogens.
3. Since there is an inactivated polio vaccine (IPV) that does not cause cases of polio, why do we still need to use OPV?
IPV provides individual protection against paralysis caused by poliovirus – wild and vaccine-derived. However, as it is an inactivated rather than a live weakened form of the virus, it does not trigger the same immune response as OPV and therefore is not effective in stopping active poliovirus transmission.
4. How important is vaccine coverage in preventing cases of vaccine-derived poliovirus?
A fully immunised population is a fully protected population. Hence, high and equitable vaccine coverage is the most important consideration when planning for the eradication of both wild and vaccine-derived polioviruses. However, the current scope and frequency of type 2 vaccine-derived poliovirus outbreaks presents particular challenges because overall population mucosal immunity against type 2 is low and continues to wane since the removal of type 2 OPV from national immunisation programmes in April 2016.
5. Did the pause in routine immunisation due to COVID-19 threaten progress against polio?
Yes. Routine immunisation coverage rates for all vaccines have in general been negatively impacted. In the case of polio, this means that there are widening immunity gaps for bivalent OPV (types 1 and 3) and IPV, thus facilitating ongoing transmission and increasing the probability of emergence of new vaccine-derived poliooutbreaks. Additionally, the COVID-19 pause was not only for routine immunisation but for outbreak response campaigns as well, further exacerbating the situation.
6. The World Health Organization (WHO) wants to transition out OPV use – is the withdrawal of OPV on schedule?
No, it is not. The gradual withdrawal of OPV started with a switch from trivalent to bivalent OPV (and the removal of type 2 poliovirus from OPV), yet this has not gone according to plan as evidenced by the spread of cVDPV type 2 outbreaks. While some cVDPV2 emergence following the switch was anticipated, particularly in the 12-18 months after the switch, the current scope and frequency of outbreaks is beyond expectations. It is now clear that different strategies and tools, i.e. vaccines – will be required to stop ongoing cVDPV2 transmission and prevent similar setbacks when eventual withdrawal of OPV1 and 3 is undertaken.
7. What is Gavi’s role in phasing out OPV and introducing IPV?
Gavi’s role has been primarily related to supporting countries in introducing IPV into routine programmes – initially a single dose and now with improved global supply – moving towards a two-dose schedule in order to improve immunity. Considerations for future OPV withdrawal will likely need to place greater emphasis on underlying routine immunisation coverage rates. Gavi’s coverage and equity agenda, and support to national programmes to strengthen delivery and coverage of children who receive no vaccinations at all (“zero-dose”) and under-vaccinated children, will be key.
8. How concerned should we be about news of cases of vaccine-derived poliovirus in Sudan?
We should be concerned by all cVDPV outbreaks. Sudan’s last reported polio case (a case of wild poliovirus) was in March 2009. Yet it has been considered at high risk for importation of polioviruses for several years, due to a decline in population immunity. With significant population movement across Sudan and neighbouring countries that are also reporting transmission of cVDPV2, the risk of further spread in and out of the country remains high. This underlines the necessity of an effective, high quality, high coverage response. More broadly, the Sudan outbreak is simply the latest in a growing number of cVDPV2 outbreaks in different regions that illustrates that the 2016 switch from trivalent to bivalent OPV has not been successful.
9. If OPV is no longer used, will IPV alone be enough, or do we actually need a new polio vaccine?
It is important to note that the removal of OPV is predicated on the eradication of wild and vaccine-derived polioviruses. Only once eradication is certified can OPV be fully removed, leaving IPV to provide continued protection to mitigate any risks related to poliovirus re-emergence. At the same time, it is increasingly clear that a new, more genetically stable live-attenuated vaccine is required. The development of novel OPV type 2 (nOPV2) has been accelerated and is in the process of obtaining Emergency Use Listing (EUL) to be used in cVDPV2 outbreaks.
10. Could vaccine-derived poliovirus threaten the eradication effort?
As long as the goal is a polio-free world – that is free of wild as well as vaccine-derived poliovirus – the emergence and spread of cVDPVs are a major setback and do threaten to at least delay achievement of a polio-free world.
We'd love to hear your feedback!
Related content
Could mRNA vaccines protect against HIV?
Moderna has begun a trial with an experimental HIV vaccine based on the same technology underpinning its COVID-19 vaccine.
Financial Times journalist and author Gillian Tett discusses her new book, Anthro-Vision: How Anthropology Can Explain Business and Life.
The Equity Agenda in Fourth Industrial Revolution Healthcare Technology
The COVID-19 pandemic has demonstrated the power of innovative Fourth Industrial Revolution (4IR) technologies in healthcare systems. Unless these technologies are designed with equity at their core, we risk leaving some people even further…
Faced with the many disease threats posed by repeated, calamitous floods, Nyando in western Kenya needs its health services more than ever. But longer-term resiliency will require more than just a shot in the arm.
Myocarditis: COVID-19 is a much bigger risk to the heart than vaccination
Myocarditis following COVID-19 vaccination is rare, and the risk is much smaller than the risks of cardiac injury linked to COVID-19 itself.
Drug-Resistant Malaria Is Emerging in Africa. Is the World Ready?
Resistance to the drug artemisinin was confirmed in Africa. Without better surveillance, it is hard to track the threat.
Endemic doesn't mean we drop our guard, surrender to the virus or downgrade the threat the virus poses to individuals or the community.
The cognitive bias that tripped us up during the pandemic
Anchoring bias meant we found it hard to get rid of the first bit of information we heard.
COVID: WHO recommends two new treatments – here’s how they work
Sotromivab and baricitinib target COVID at different stages of its development, and will give doctors greater flexibility in treating Omicron patients.
"A big win-win": Vaccine sharing could protect against future COVID-19 waves
Modelling suggests that increased COVID-19 vaccine donations by wealthy nations could benefit rich and poor countries alike.
Q&A: Religious minorities and ‘faith’ in vaccines
Nurturing confidence in vaccination among minority populations like the Haredim, a Jewish religious group sometimes problematically referred to as “ultra-Orthodox,” has a lot to do with listening, says medical anthropologist Ben Kasstan.
How the planes of the future will create a healthier planet
The effects of climate change may lead to future pandemics. As the aviation industry looks to the future, cleaner fuels and electric planes will be key to reducing aviation’s impact on the climate.
Genetics helps estimate the risk of disease – but how much does it really tell us?
How to make sense of 'polygenic' risk scores.
How a private-public partnership is training supply chain professionals in the global south
‘Soft skills’, from motivating a team to tapping into networks, provide the vital oil that keeps supply chains moving smoothly. A programme called STEP is bringing together businesses and the public sector to help healthcare managers in the…
Mallory Smith died at 25 with cystic fibrosis, of an antibiotic-resistant bacterial infection. Amid rising rates of AMR, the elegiac sadness of her memoir-in-film feels like a warning.
COVID: why some people with symptoms don’t get tested
Recent research found around a quarter of people with classic COVID symptoms didn't get tested.
We now know a major cause of multiple sclerosis. Can we eradicate it?
A new study shows that Epstein-Barr virus infection is strongly linked to multiple sclerosis. There is currently no cure to multiple sclerosis, and access to innovative treatments is limited in many countries. Vaccinating for EBV instead could…
How unequal access to vaccines could threaten climate negotiations
Wealthy countries have given their citizens third booster doses, yet have so far failed to ensure that those in less well-resourced nations are also protected; experts warn this inability to respond to a shared crisis could threaten climate…
We can stop the cycle of new variants continuing indefinitely with a pandemic vaccine pool
Without it we could face further delays in the global rollout and risk the gulf between the vaccine haves and have nots widening.
Antimicrobial resistance now causes more deaths than HIV/AIDS and malaria worldwide – new study
Latest numbers show antimicrobial resistant infections contributed to 4.95 million deaths globally in 2019.
COVAX and the other international organizations committed to vaccine equity cannot end the COVID-19 pandemic without the continued support of governments, industry, and civil society. By working together, the world has a chance to tame the…
Here’s where (and how) you are most likely to catch COVID – new study
To discover your risk of catching coronavirus for any given situation, try our COVID-19 Aerosol Transmission Estimator.
Scientists around the world are working faster than ever to develop and produce vaccines that can stop the spread of COVID-19, with 21 vaccines now being rolled out in countries worldwide. Here is an at-a-glance overview of those vaccines and…
Omicron: Vaccines remain the best defence against this COVID-19 variant and others
Even with a variant like Omicron that might be more transmissible than earlier variants, vaccines remain the most effective tool for protection against COVID-19 and for ending the pandemic.
How COVID-19 transformed genomics and changed the handling of disease outbreaks forever
Revolutions in genome sequencing have been used to track COVID-19 in near real time.
Churches are boosting vaccination rates in Eswatini
Eswatini has launched a campaign to encourage members of the Zionist churches to get vaccinated against COVID-19.
COVID healthcare costs ‘catastrophic’ for poorest
Health care costs push more than half a billion people into extreme poverty. COVID-19 adds to the burden, halting progress towards universal health coverage. Health policies must protect poor from financial hardship, say World Bank, WHO.
Long Covid is Pitting Patients Against Doctors. That’s A Problem.
Health care professionals must act swiftly to ensure frustrated patients don’t fall victim to misinformation.
All around the world, COVAX deliveries are helping to protect frontline health workers, reach nomadic communities, reunite families and allow schools to reopen. Since deliveries began in March, #VaccinesWork has been publishing snapshots that…
Oral Cholera Vaccine saving lives in South Sudan
While South Sudan has been declared cholera-free, it is an ongoing battle to keep things that way.
Cholera’s 7th Pandemic turns 60
During the six decades of its 7th pandemic, cholera has become an avoidable, survivable scourge. But instead of ebbing away, the disease has emerged more starkly than ever as a vulture of inequity.
Q&A: “We have the momentum” – fighting to end a 60 year-old cholera pandemic
This year, the ongoing seventh cholera pandemic turned 60. Here, Lorenzo Pezzol, epidemiologist and cholera expert, tells #VaccinesWork that we have the tools to end it – and that we really should have done so long ago.
#VaccinesWork’s top 11 articles of 2021
2021 has been another tumultuous pandemic year. #VaccinesWork has been there through every twist and turn in our collective pandemic story, covering the latest science, bringing new perspectives from countries across the world and digging into…
Greetings from the North Pole to the Polarised North: street art Santa arrives in Germany
An expression of frustration with vaccine skeptics, German street artist and former immunologist Lapiz's new work is not your typical Christmas card.
Dr Seth Berkley's reflections on 2021 and the global imperative for 2022
2021 has been another challenging pandemic year, but despite the challenges it saw important milestones, extraordinary resilience in Gavi-supported countries and progress on a number of fronts. In his annual end of year letter, Gavi CEO Dr Seth…
How COVID-19 and Omicron are affecting travel this holiday season
Here's what you need to know about Omicron and traveling safely this holiday season.
Why excess deaths have varied so greatly around the world during the pandemic
How governments chose to respond to the coronavirus – and how well equipped their health services were before it arrived – made a big difference.
Tackling COVID disinformation with empathy and conversation
Living with someone who believes in anti-vaccination misinformation can tear families apart, say experts.
The Pandemic and Africa's Social Safety Net
The COVID-19 pandemic has shown that African tax and social-benefit systems are currently ill-equipped to protect households from sudden income losses. Meaningful progress will require policymakers to reduce the size of the informal sector and…
An ode to health workers on the highways of Monterrey, Mexico
Street artist Buffon draws attention to the importance of healthcare workers and how we can all play our part in ending the pandemic.
This is how COVID-19 hit household expenditure in Europe
New data reveals that COVID-19 reduced Europe’s household spending by 8% in 2020. Nations with the severest lockdowns saw the most dramatic drop in household consumption, says Eurostat. Elsewhere, consumer spending in China fell by 17% in 2020.…
Galápagos census looks at impacts on turtles during and after COVID lockdown
The suspension of tourism activities around the world as a result of the COVID-19 pandemic gave researchers the chance to answer an important question: What impact does tourism have on wildlife populations?
The groundbreaking history of the world’s first malaria vaccine
Malaria occupies a unique place in human history. Our neolithic ancestors were afflicted by it; the fall of Rome has even been partially attributed to it. During the 20th Century alone, malaria is estimated to accounted for up to five percent of…
"The vaccine has done wonders”: Kenya’s experience with the world’s first malaria vaccine
The Gavi Board has approved the roll-out of RTS,S/AS01, the world’s first vaccine against malaria, a sickness which kills 260,000 children under five in sub-Saharan Africa every year. To get a sense of what this historic development might mean on…
When will life return to normal after the pandemic?
Surveying by the Office for National Statistics shows Brits are increasingly pessimistic about things returning to how they were before.
Growing viral: health, hope, and happiness in the time of COVID and HIV
Living with one lethal virus, the children of Snehagram in southern India bolster each other in surviving the coronavirus pandemic.
Mask wearing wasn’t disputed in previous crises – so why is it so hotly contested today?
How the pandemic is reported by the media can influence people's behaviour.
The hunt for coronavirus variants: how the new one was found and what we know so far
There's a new COVID lineage called B.1.1.529. It has a genetic profile very different from other circulating variants.
There's Only One Way to Stop the Emergence of Variants Like Omicron
"This crisis is far from over, and without dramatic course correction, will go on for some time."
Omicron: why the WHO designated it a variant of concern
Not much data, but lessons have been learned from the slow response to delta.
Measles is one of the world’s most contagious viruses. It can impair sight, hearing and cause brain inflammation and pneumonia. But even though it can be prevented by a vaccine, outbreaks in unvaccinated children continue to erupt even in the…
How to gauge a plague: new analysis sheds light on the meaning of a 6th century pandemic
Disease outbreaks have shaped the world. But how much? New thinking on an early medieval pandemic takes on “plague sceptics” to show that we likely underestimated the impact of the first cross-continental assault of bubonic plague.
Maternal vaccine urgently needed to protect babies against Strep B
A new report reveals the devastating global impact of Group B streptococcus, but suggests vaccinating against it could be highly cost-effective – if an effective vaccine can be developed.
Study: government vaccine spending increases since 2000
A comprehensive analysis of vaccine spending shows that while lower-income country governments take on the bulk of financing, they need to mobilise greater resources for immunisation.
COVID-19 and the impact on part time jobs
The loss of jobs during the pandemic was experienced almost exclusively among part-time jobs. Research shows it is likely that demand for workers in hard-hit sectors will start to come back, writes economist Jonathan Wadsworth. However, the…
Why COVID-19 Misinformation Works
The rampant spread of false information about the coronavirus has been attributed to politicians who have promoted remedies ranging from anti-malarial drugs to herbal drinks. But how humans process knowledge has an important role to play in…
How Peru became the country with the highest COVID death rate in the world
The country moved quickly to contain the virus, but its health system struggled to look after those who got sick.
Young and ethnic minority workers were hardest hit at the start of COVID, but not anymore
The two groups bounced back more quickly in the jobs market than certain other groups, although they may still experience lasting consequences.
Hospital waste, not masks, are plastic scourge of pandemic: Study
A new study has found that 26,000 metric tons of pandemic-related plastic waste has been released into the world’s oceans since the start of the COVID-19 pandemic in January 2020.
Luciferase is a useful tool in medicine and has nothing to do with Satan.
Is this the great vaccine-mandate resignation that wasn’t?
COVID-19 vaccine mandates have spurred protests and resignations. But the evidence so far points to marginal workforce losses. Evidence also suggests that mandates keep people safe and save lives.
My shampoo stinks of vomit: How COVID-19 can mess up your sense of smell
Increasingly, people who are recovering from COVID-19 are reporting a distorted perception of odours, also known as parosmia.
Why having bad oral health could raise the risk of COVID
Badly controlled bacteria in the mouth pose multiple risks.
How disease has stimulated cultural change
Laws and rituals surrounding disease have been part of everyday life for millennia. Here's why that's important.
History’s Seven Deadliest Plagues
SARS-CoV-2 has officially claimed 5 million lives, but credible estimates place the pandemic’s true death toll closer to 17 million. Either count secures COVID-19’s position on our list of history’s deadliest plagues.
What pneumonia can teach us about COVID-19
COVID-19 still has many unknowns, but hope lies in what we already know about preventing and treating pneumonia.
Two-strike Ebola vaccine enters human trials
The vaccine, which is based on the Oxford/AstraZeneca COVID-19 vaccine, is designed to target both the Zaire and Sudan species of Ebola virus.
India’s pneumococcal vaccine rollout will save 50,000 children a year
Pneumococcal Conjugate Vaccine has now been introduced in every state in India, meaning this lifesaver will be available to every child in the country.
Investments in Disease Surveillance Support Kenya’s Response to COVID-19
In a conversation with Maria Deloria Knoll, PhD, IVAC’s Director of Epidemiology, Dr. Kagucia shared how Gavi-supported activities facilitated Kenya’s COVID-19 response by expanding capacity, testing, and partnerships.
Can vaccines unlock access to health care?
Zero-dose children who never receive a single vaccine are also more likely to miss out on crucial primary health care services.
Sickness and survival in pre-COVID-19 Nepal
A gripping documentary about a family's struggle with the emotional and financial consequences of severe pneumonia.
Nose sprays, needle-free patches, durable immunity: towards the next generation of COVID vaccines
The development of COVID vaccines has already been explosive. There are more innovations on the way.
White-tailed deer found to be huge reservoir of coronavirus infection
In a sample of white-tailed deer, 80% were found to have an active COVID infection.
HPV vaccine cuts cervical cancer cases by nearly 90%
Real world data from the UK shows the value of vaccinating girls against human papilloma virus (HPV) when they are 12 to 13 years of age.
Dr Thomas Tu has an intimate relationship with the virus he’s devoted his life to studying, having been diagnosed with chronic hepatitis B at the age of 14. He discusses the stigma associated with the disease and the future of treating it.
Five crucial years, one unprecedented new pandemic and 324 million children immunised
Even with the adversities and challenges of the COVID-19 pandemic through 2020, Gavi maintained unyielding focus on leaving no one behind.
Hepatitis B kills more people each year than AIDS-related illnesses, yet an effective vaccine exists. Ensuring every child has access to it is crucial to fighting back.
Vaccine profiles: Haemophilus influenzae type b (Hib)
Before immunisation for Hib became routine, it was one of the biggest killers of children under five and the main cause of meningitis. High vaccine coverage is essential to keep that threat at bay, especially given rising antimicrobial resistance…
COVID could wreak havoc on gorillas, but they social distance better than we do
Endangered mountain gorillas in Rwanda’s famous Volcanoes National Park could face “population collapse” within 50 years if some of them contract COVID-19, new research suggests.
What COVID-19 is teaching us about human milk and antibodies
Dr Rebecca Powell is on a quest to understand the immune response to infection and vaccination in breastmilk, in the hope of designing maternal vaccines to boost babies’ protection against disease.
The walls speak for the vulnerable in Luanda
Oksanna Dias has been using street art to draw attention to the challenges of the women of Luanda.
Injectable rotavirus vaccines – what value could they bring?
Currently in advanced development, these next-generation rotavirus vaccines may address key barriers to vaccine uptake and access.
The disease can kill one in five people infected, yet an effective vaccine exists. Routine tetanus immunisation saves the lives of thousands of newborns every year.
Are COVID-19 vaccines safe for teenagers and children?
Some countries are already offering COVID-19 vaccines to teenagers and may expand this to children in the near future. Here’s what we know about vaccine safety and efficacy in these groups.
COVID: how worried should we be about the new AY.4.2 lineage of the coronavirus?
This new sub-variant of the delta has gained a foothold in the UK.
Before immunisation for pertussis, or whooping cough, became routine, it killed twice as many children as measles and polio combined. High vaccine coverage is essential to keep that threat at bay.
Women Leaders in Polio Eradication: Dr. Folake Olayinka
Dr. Folake Olayinka speaks about overcoming gender-related barriers to immunization.
As India celebrated its one-billionth COVID-19 vaccination, #VaccinesWork caught up with Anuradha Gupta, Gavi’s Deputy CEO and an Indian public health veteran.
Is it a cold or COVID-19? An expert explains
Research in the UK shows COVID-19 symptoms can be similar to the common cold. So, how can you tell the difference? Here, genetic epidemiologist Professor Tim Spector explains the latest data and what you need to know.
Before immunisation for diphtheria became routine, it was a major killer of children and adults worldwide. High vaccine coverage is essential to keep that threat at bay.
COVID and flu: how big could the dual threat be this winter?
Social distancing has shielded us from other respiratory viruses – but that's not necessarily a good thing.
First flight facilitated by the Hope Consortium touches down in Belgium with large delivery of ultra-cold chain freezers for onward shipment to 21 African countries.
Identifying people who have recovered from COVID-19 could provide a way to accelerate protection of populations against the disease.
Without women there are no healthcare systems
The sixth annual Heroines of Health Gala celebrated seven women fighting to improve the health of their communities. From mobilising nurses in Beirut after the blast in August last year to helping communities in Ethiopia affected by the civil…
How COVID-19 is contributing to period poverty in Nigeria
Skyrocketing prices for sanitary products, supply shortages and lockdowns are combining to make it harder and harder for girls and women in Nigeria to manage their periods.
Breakthrough malaria vaccine offers to reinvigorate the fight against the disease
The WHO and the manufacturers of the vaccine will be rallying countries, particularly those with high malaria burdens, to adopt the vaccine.
Drought may affect routine immunisation in Africa, study finds
Climate change is set to transform global health. A new study has found that droughts – one product of extreme weather exacerbated by climate change – may already be impacting rates of vaccination.
Kenya is aiming to eliminate malaria entirely by 2030, with the world’s first malaria vaccine forming an important tool in its arsenal. The country’s youth is playing an important role.
First ever malaria vaccine recommended by WHO: Q&A with Deepali Patel
The WHO recommendation on the RTS,S vaccine could protect millions of children across Africa.
Nobel Prize: how chili peppers helped researchers uncover how humans feel pain
These discoveries could help us treat a variety of conditions in the future – including chronic pain.
Further vaccine R&D is critical to end the devastating COVID-19 pandemic
It is science that we must once again turn to so that we can remain one step ahead of the virus. Increased investment in ongoing vaccine R&D efforts will enable COVAX to deliver on its promises and ensure that our vaccines remain safe and…
Head of UNAIDS unpacks the knock-on effects of COVID-19. And what needs to be done
Greater urgency is needed in the response to the pandemic, to end AIDS and to end COVID-19.
Smokers were never really protected from COVID, despite what early studies claimed
How the 'smoker's paradox' went up in a puff of smoke.
The COVID-19 vaccine-shy are changing their minds
Vaccine hesitancy is “not a stable trait,” concludes a new American study, which found that a majority of initially hesitant respondents shifted their stance in favour of getting the jab.
How COVID-19-related stress could be affecting your menstrual cycle
A new study suggests that more than half of women may have experienced irregularities in their periods after the COVID-19 pandemic began.
Routine immunisation worldwide holds firm despite the pandemic
Despite the havoc wrought by the COVID-19 pandemic on health systems worldwide, governments’ work to bring immunisation programmes back on track is paying off.
COVID-19 vaccines help routine immunisation become routine again in Nigeria
Routine immunisation took a hit in Nigeria thanks to the pandemic. Thanks to the arrival of COVID-19 vaccines, particularly for health workers, mothers are returning to immunisation clinics.
Vaccinating domestic dogs reduces rabies in the wild. Why this matters
Domestic dogs have been shown to be the only species necessary to maintain rabies across most of Africa. This means that dog vaccination should control the disease in all species.
Whipping the Covid-19 vaccine market into shape
The COVAX vaccine procurement facility has run a remarkable race, but needs stable funding for long-term success.
Your immune system is as unique as your fingerprint – new study
New discovery could help scientists develop more targeted drugs and vaccines.
Cholera and the inequitable origins of public health diplomacy
The arrival of cholera on European shores in the 19th Century helped kick off the long path towards modern global health institutions and diplomacy. However, these beginnings were anything but fair.
Lower-income countries are letting far fewer COVID-19 vaccine doses expire than wealthier nations
Just 0.2% of COVID-19 vaccine doses delivered to lower-income countries through COVAX expired before they could be administered, estimated to be a significantly lower amount than wealthier countries.
The historic push to provide ultra-cold chain freezers around the world
Thomas Sorenson, Senior Manager in the Cold Chain Unit, UNICEF Supply Division, explains the work behind the scenes to roll-out the largest ultra-cold chain deployment in history.
This is the WHO’s massive new list of ways to create a healthier, safer environment
Four UN agencies have compiled a first-of-its-kind Compendium of guidance on health and environment to help policymakers save millions of lives. The Compendium pulls together 500 actions and recommendations on everything from clean water and…
Reaching zero-dose children in Malawi’s cities
In Malawi urban areas have more unimmunised zero-dose children than rural regions. The government is working hard to close the immunisation gap.
Gavi at UNGA: Action for equity
The 76th UN General Assembly (UNGA 76) will take place from 14-30 September 2021. Could we see a breakthrough in the fight for universal vaccine access?
Even a quarter dose of Moderna vaccine triggers strong immune response
Although it’s not clear how this compares to the full dose licensed worldwide for emergency use, a quarter of a dose produces an equivalent immune response to immunity from infection.
Now we know: typhoid conjugate vaccines are highly effective in African children
A study in Malawi found that TCVs are 84% efficacious among children 6 months to 15 years of age – the first such evidence among African children.
Q&A: Helping teenage mothers access immunisation in Rwanda
The COVID-19 pandemic has reportedly increased the number of teenage mothers across Africa. Inadequate childcare support and stigma can prevent teenage mothers accessing routine immunisation and nutritional support for their infants. Girl Effect…
COVAX: the forecast for vaccine supply
While the supply is lower than predictions earlier this year, wealthy countries and manufacturers hold the power to improve supply.
You don’t need to worry about COVID vaccines being ‘unnatural’ or ‘synthetic’
mRNA vaccines are the first synthetic vaccines, meaning they are made outside of a living cell. But so are lots of things we consume every day, such as vitamin C pills and other dietary supplements.
Review: Survivors – Hope and Resilience in the Time of Ebola
The Sierra Leonean documentary is a complex portrait of courageous solidarity amid contagion.
Pop culture icons and political satire in Bristol
The British artist John D’oh uses humour and wit to drive home messages about vaccines and other measures such as hand-washing and social distancing to stop the spread of COVID-19.
We’re getting closer to a vaccine against Chikungunya
Launch of vaccine trial marks “significant milestone” towards beating this debilitating disease.
How improved yellow fever diagnostics are transforming management of the disease
Gavi has supported a major expansion in yellow fever diagnostic capacity in Africa over the past three years. The results show just how much improving diagnostics can have a cost-effective yet significant impact on immunisation programmes.
Anand Kumar: From polio to para-sports world champ
More often than not, the road to a meaningful triumph is a bumpy one.
The G20 Must Recommit to COVAX
Additional G20 support can make the COVID-19 Vaccine Global Access facility a success. Failure to achieve equitable access would mean more lives lost, broken health-care systems, even deadlier and more transmissible variants, and a pandemic with…
Protecting a different kind of frontline worker in Liberia
Getting market women and pen-pen drivers vaccinated is critical to tackling COVID-19 in Liberia.
Why helping countries deliver COVID-19 vaccines is just as vital as boosting supply
Getting COVID-19 vaccines out to countries isn’t just about ensuring supply – a major stumbling block can be their ‘absorptive capacity,’ or the ability to deliver the volume of vaccine coming their way. Here, we talk to Alex de Jonquieres,…
COVID-19 boosted excess sale of antibiotics in India
COVID-19 boosted sales of antibiotic formulations used in adults and adolescents in India. Antibiotics should not be prescribed to treat viral diseases. Misuse of antibiotics can result in drug-resistant infections.
Hantavirus study shows restoring forests can reduce zoonotic disease risk
Brazilian scientists have found that transmission of hantavirus, a deadly infection, could be reduced by 45% if Brazil restores its Atlantic Forest to levels mandated by its Native Vegetation Protection Law. Hantavirus, spresd by contact with…
The first human case of Marburg virus in West Africa is no surprise: here’s why
The Marburg virus will be present wherever the Egyptian Rousette bat occurs.
Humanitarians Push to Vaccinate in Conflict Zones
Pandemic ceasefires offer an opportunity to expand vaccination efforts, experts say. But negotiation is tricky.
COVID variants: we spoke to the experts designing a single vaccine to defeat them all
A universal vaccine has been described as the ‘holy grail’ – but how close are we to getting one?
Do kids get long COVID? And how often? A paediatrician looks at the data
Children can get long COVID, but it seems to be less common than in adults. And they tend to recover quicker.
More than a quarter of a million infants estimated to have died due to pandemic poverty
More than a quarter of a million infants in low- and middle-income countries are predicted to have died in 2020 due to the economic impact of COVID-19.
With evidence showing the COVID virus is airborne, it's no coincidence many outbreaks in schools have occurred in winter – when windows are closed.
With COVID-19 stretching the healthcare system in many parts of Nigeria, health workers and community leaders have to go the extra mile to ensure vaccines reach victims of the country’s latest cholera outbreak.
A new study suggests the RTS,S malaria vaccine alone is as effective as preventive antimalarials; together they could save thousands of lives.
In Brazil’s Acre, smoke from fires threatens health, could worsen COVID-19
Wildfire smoke has been linked to higher COVID-19 mortality rates, threatening to compound what is already one of the worst burdens of coronavirus infections and deaths in the world. At particular risk are Indigenous populations, who suffer…
After India’s brutal coronavirus wave, two-thirds of population has been exposed to SARS-CoV2
A public health scholar based in New Delhi explains how India has emerged from the massive spike in COVID-19 cases, even as the country braces for a new wave.
Are COVID-19 vaccine mandates human rights violations?
Experts point to a more fundamental right to be protected from the disease, particularly as it continues to spread primarily among the unvaccinated.
Why I no longer think we can eliminate COVID – public health expert
As New Zealand enters another lockdown after detecting a single COVID case, it's time to consider whether stringent zero COVID strategies are feasible in the long term.
In the absence of good vaccines, doctors treating epidemic diseases must rely on cumbersome PPE as their one safety net. At least modern hazmat suits, unlike the 17th century plague doctor's all-leather outfit, actually work.
How to eliminate cervical cancer - an expert explains
COVID-19 has caused a decline in the delivery of human papillomavirus (HPV) vaccines that protect girls from cervical cancer. Anuradha Gupta, the Deputy CEO of Gavi, explains what we must do to create more access to these vaccines.
Vaccines can win the race against COVID-19 variants. Here's how
COVID-19 variants threaten to undermine global vaccine programmes unless we accelerate the pace of delivery and rise to the challenge of global deployment.
Should we tell stories of vaccine sceptics who have died of COVID?
The news is full of stories of people who refused the jab who died of COVID.
Vaccines bring hope for children in Nigeria’s largest coastal slum
Access to routine immunisation and primary health care is changing the lives of children in Makoko, Nigeria’s largest coastal slum.
Investing in Global Vaccine Equity Acknowledges Our Shared Fate
Vaccines are among modern medicine’s greatest innovations, allowing billions of people to lead healthy lives. But stopping outbreaks of vaccine-preventable disease – and not only COVID-19 – depends on achieving critical mass with immunization…
Plagues and classical history – what the humanities will tell us about COVID in years to come
We all need to know about the science of COVID as we battle through pandemic, but the ultimate story will lie in how it changed our society.
TCV is 85% efficacious and urgently needed for Bangladeshi children
New study finds TCV prevents 85% of cases among vaccinated children in Bangladesh, which has one of the highest typhoid burdens in the world.
How bad will climate change be for our health?
The latest IPCC report emphasises the inevitability of some degree of climate change. The human health impact of this should not be ignored.
John Snow and the Pump Handle of Public Health
The narrative of the Broad Street pump is the story of a vital epidemiological discovery; it’s also a story about discovery: it has the tidy, conclusive quality of a fable about science. But in the years after its discovery, John Snow’s theory of…
RSV: what is it, and why are child cases surging in the wake of COVID?
COVID restrictions have prevented the spread of other respiratory viruses too. As these are lifted, the UK government has warned parents of a delayed surge in infections.
That Old Time Anti-Vaxx Feeling
The best single predictor of vaccine uptake per US state is not political affiliation, but the share of the population that believes the human race has always existed. Such findings do not bode well for the global effort to boost vaccination…
COVID: why you still need to wash your hands
Respiratory viruses, such as influenza and SARS-CoV-2, while spread primarily by respiratory droplets can also be spread by contaminated hands touching the mucous membranes of the nose, mouth and eyes.
In 2014, Nigeria escaped an Ebola pandemic in the span of 93 days because of the bravery and quick response of health care workers in Lagos. As the world fights through the COVID-19 pandemic, the film 93 Days – though grim – is a hopeful reminder…
Reaching out to communities in rural Zambia
In rural Zambia, clinics and health centres rely on a limited number of medical personnel to run immunisation programmes, which makes the support of community leaders vital.
Review: The Cutter Incident by Paul Offit
Offit’s sobering inquiry into “one of the worst biological disasters in American history” is a work of unflinching investigative journalism which lands, finally, not as a whodunit, but as a tribute to modern systems of meticulous vaccine…
How do experts ensure vaccine safety?
From clinical trials to population surveillance, these are the multilevel mechanisms that ensure vaccine safety – including the safety of COVID-19 vaccines.
Intellectual Property and COVID-19 vaccines
During a pandemic supply bottlenecks of essential medical products are almost inevitable, but with COVID-19 this has been further exacerbated by governments hoarding doses and imposing export bans.
A Deadly Alliance – War and the Pandemic Influenza of 1918
As World War I reached its climax, a terrible influenza pandemic broke out. By summer 1919, it had claimed many more lives than the conflict – but the conflict, researchers say, helped create the conditions for the devastating spread of the so-…
“My heart is always stuck in my homeland”: A Rohingya returnee’s story
Abdullah and his family escaped genocidal violence in Myanmar in 2017, survived a diphtheria epidemic in Bangladesh’s Cox’s Bazar and weathered the first year of the COVID-19 pandemic in a Rohingya camp. Then, in December 2020, he returned to the…
“Saving the future of our children”: The women fighting polio in Pakistan
Pakistan is one of two countries where wild poliovirus remains endemic making it essential that the entire country supports the successful implementation of every polio campaign. Women play a critical role, often working at polio’s frontline.
COVID masks: how and when to ask someone to wear one – without getting into a fight
Just because face coverings aren't mandatory doesn't mean they're not protective – so here's how to ask someone to wear one.
Review: COVID, Quickly – A Scientific American podcast series
This series cuts through the noise to help you catch up with the latest COVID-19 developments in an entertaining and digestible way.
TB prevention has relied on the same vaccine for 100 years. It’s time for innovation
BCG remains the only widely available vaccine for TB. Yet the development of a COVID-19 vaccine over the last year shows that there is capacity to rapidly create new vaccines.
How effective are COVID-19 vaccines in the real-world?
Real-world data is starting shed light on the performance of COVID-19 vaccines in less than perfect conditions.
Q&A: Vaccine – The Human Story
Dr Annie Kelly, writer and host of a new podcast which tells the story of the smallpox vaccine, talks to #VaccinesWork.
Review: The Panic Virus by Seth Mnookin
Mnookin’s 2011 investigation of the spread of anti-vax feeling is a pandemic-era must-read.
New vaccination figures underscore urgency to reach the most vulnerable
New data shows the impact COVID-19 is having on routine immunisation programmes worldwide, particularly in lower-income countries. We need action, now, to get routine immunisation programmes back on track and ensure that everyone, everywhere is…
Rather than translating their own COVID-19 vaccination successes into a renewed global push to end the pandemic, rich countries are becoming complacent while the rest of the world grows increasingly desperate. But the emergence of dangerous new…
How investigation into long COVID will help us create treatments.
Willing and waiting: High levels of COVID-19 vaccine acceptance identified in Global South
The widespread acceptance of COVID-19 vaccines in low- to middle-income countries should boost global coronavirus immunity, if distribution issues can be resolved.
Why Africa needs to manufacture its own vaccines
Dr John Nkengasong is a Cameroonian virologist and Director of the Africa Centres for Disease Control and Prevention (Africa CDC), which is working to strengthen the ability of Africa’s public health institutions to detect and respond to disease…
COVID: should you ditch your mask once restrictions are lifted? A philosopher’s view
A philosopher of public health ponders whether taking your mask off on July 19 is like punching someone in the face or like riding a bike.
How Do We Track Vaccinations for People Who Don’t Formally Exist?
Yoshinobu Nagamine, Senior Donor Manager at Gavi, the Vaccine Alliance, describes the role contactless biometrics can play in accelerating vaccine delivery for those lacking formal identification.
COVID-19: how a sense of community can increase vaccine uptake
If people feel a sense of duty to their local community, they're more likely to protect themselves and others by taking the COVID-19 vaccine.
We should treat COVID like norovirus – not the flu
Comparisons with flu are unhelpful – these diseases need to be treated differently.
Pen and paper in a digital age: tracking childhood immunisation in Kenya
It may not be high tech, but it works. Kenyan community health volunteers use tracking books to ensure that children receive routine vaccinations.
COVID: lambda variant is now in 29 countries, but what evidence do we have that it’s more dangerous?
The lambda variant ripped through Peru and is now spreading globally. But there is little evidence that it is more dangerous than the original variant.
Could the seasonal flu vaccine help protect against COVID-19?
A large study suggests the influenza vaccine may reduce the risk of some of the worst consequences of COVID-19.
COVID-19 vaccine boosters: is a third dose really needed?
If immunity is waning or ineffectual against new variants, then boosters could be needed – but there needs to be evidence that they're necessary.
Why Africa’s push to make vaccines should look further than COVID-19
Vaccine manufacturing doesn't come cheap. It depends heavily on support from developed countries. It also takes much more than relaxing intellectual property rights and a desire for vaccine equity.
Handwashing ‘impossible’ for one in three amid COVID-19
One in three unable to properly wash hands at home during COVID-19 – WHO-UNICEF. Progress on water, sanitation and hygiene ‘must quadruple’ to meet 2030 target. Investment and prioritisation needed ‘at the highest levels’.
Is COVID-19 effectively already endemic?
Will the pandemic go out with a fizzle or a bang? And what does “living with the virus” actually mean, and when will we get there?
Opinion: Reach 'zero-dose' children to build back better
We must seize this opportunity to shape our COVID-19 response as a chance to achieve better equity and social justice. What better way to do that than to focus on zero-dose children?
COVID variants: could dangerous new ones evolve in pets and farm animals?
Early results from several studies have found that pets can pick up COVID-19 from their owner – but they are unlikely to be dangerous as a result.
Africa ‘tech hub’ aims to fill COVID-19 vaccine gap
New mRNA tech transfer hub to be established in South Africa. Facility will provide training and licences for vaccine development. Less than one per cent in Africa have been vaccinated against COVID-19.
The power sources used to keep vaccines cold used to be polluting and unreliable. We spoke to Alex de Jonquieres, Gavi’s Director of Health Systems and Immunisation Strengthening about how Gavi, the Vaccine Alliance, became investors in solar…
How Can I Spot A Fake COVID-19 Vaccine?
Fake COVID-19 vaccines erode public trust in much-needed jabs and encourage vaccine hesitancy.
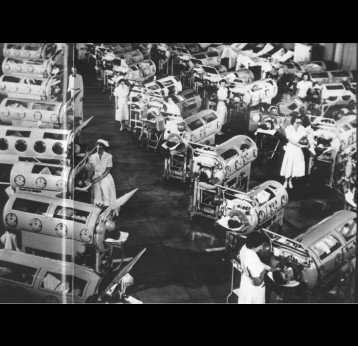
Before 1955, when a vaccine first made polio a preventable illness, the paralysing disease had to be treated. For many, the best option was the iron lung, a device that came to symbolise an era of anxiety in mid-20th century America.
Why sharing bioimaging tech is crucial to global health
New bioimaging technologies are pushing the boundaries of medicine. But poorer nations risk losing out on these advances. Investment in facilities and training globally is crucial to bridge the divide.
Are men and obese people really at greater risk of dying from COVID-19?
A reanalysis of 58 existing studies suggests that with COVID-19 being male or seriously overweight isn’t as risky as originally thought.
Guinea declares Ebola epidemic over
Guinea declares end of Ebola outbreak after 42 days without new cases. Outbreak resulted in 23 likely cases, 12 deaths since February. Success attributed to community buy-in, capacity building and vaccination.
What you need to know about COVID-19 vaccines and myocarditis in teens
This rare side-effect from the two RNA vaccines being rolled out might be concerning but the benefits of vaccination far outweigh the risks.
COVID-19 vaccines: why it’s important you get your second dose
Two doses have always been more protective than one, but the Delta variant has made the benefit of the second dose much greater.
The Immunization Agenda 2030’s new scorecard visualizes a journey toward pandemic recovery and global immunization progress.
I’m fully vaccinated but feel sick – should I get tested for COVID-19?
Vaccinated people can still get infected with the coronavirus. So if you have symptoms of COVID-19, getting tested can protect others and help health officials keep an eye on the virus.
The COVID-19 pandemic has affected turn out for almost all Malawi’s health services, including routine immunisation. One local CSO is using Community Mother Care groups to trace those missing out on vaccines, helping to ensure children are…
New systems map can help overcome difficulties in vaccinating hard-to-reach communities
A major obstacle for COVID-19 vaccination programs to achieve higher and more equitable vaccine coverage throughout the world is vaccinating hard-to-reach communities.
What does COVAX’s latest supply forecast tell us?
The forecast is encouraging given the supply disruptions COVAX and countries with bilateral deals are currently experiencing, and the general challenges manufacturers are experiencing as they ramp up supply at historic speed and scale.
We archived 84 million tweets to learn about the pandemic – each one is a tiny historical document
During the pandemic, researchers have treated Twitter as a sprawling and evolving historical document.
Giving 110%: Eswatini’s early rollout of COVID-19 vaccines
Eswatini received its first batch of 32 000 Oxford-AstraZeneca vaccines in late March. 12 000 came through the COVAX facility and 20,000 through a donation from the Indian Government.
Q&A with Sterling and Seth: Spotlight on COVAX
What do the CEO of the Vaccine Alliance and the star of a hit US TV show have in common? They both want to raise awareness about COVID-19 vaccines and COVAX’s role in getting the world vaccinated.
Mary Mallon was branded a public menace and incarcerated after typhoid outbreaks across New York City were linked to her presence in the kitchen. Was she unfairly demonised?
Needle phobia could be the cause of 10% of COVID vaccine hesitancy in the UK – new research
People who are afraid of needles are twice as likely to be vaccine hesitant, new research shows.
Does a plant-based diet really help beat COVID-19?
A new paper suggests that plant- and fish-based diets lessen the chance of developing severe symptoms – but hold off from becoming vegetarian or pescatarian for now.
From the great plague to the 1918 flu, history shows that disease outbreaks make inequality worse
Accounts of previous epidemics – by Samuel Pepys, Daniel Defoe and Katherine Porter – warn of mistakes that we risk repeating.
Tackling typhoid in Zimbabwe: “We have to be sure for ourselves that the water is safe”
After a devastating typhoid outbreak swept through Bulawayo, Zimbabwe, a new, more effective vaccine is bringing hope.
To keep coronavirus at bay, we must create a ‘variant-proof’ world
We remain locked in a deadly race with the virus. We will only win when we have created a “variant-proof” world that can keep a lid on the havoc caused by its troublesome mutations.
The ‘black fungus’ epidemic hitting COVID-19 patients in India
Amid the 28.4 million cases and over 300 thousand deaths from SARS-CoV-2, a second epidemic has hit India. ‘Black fungus’, clinically known as mucormycosis, is an infection currently affecting nearly 12,000 immunocompromised patients recovering…
How likely is a positive COVID-19 lateral flow test to be wrong?
Rapid antigen tests for COVID-19 are less sensitive than PCR tests, but here we explain why you should never ignore a positive result.
Is 150 years really the limit of human lifespan?
Researchers think they've calculated the limit of human lifespan – but there's more to it.
Speeding up vaccine development: Can we go from lab to jab in just 100 days?
COVID-19 vaccines were made in record speed, taking around 300 days from the moment the threat was first identified. But the world’s top scientists are aiming to overtake this world record in the next pandemic, aiming to make a vaccine in 100…
COVID-19's impacts on the brain and mind are varied and common – new research
Non-specific illnesses, such as headache and fatigue, are common, as are mental illnesses such as depression and anxiety.
LGBTIQ people have been hit hard by COVID-19. Here’s how we can all help
Marginalized people suffer from being at the intersection of multiple challenges and barriers. Economic hardship has been a secondary epidemic in the LGBTIQ community. We need public health and support services that acknowledge difference.
Five reasons why the real number of COVID-19 deaths could be triple the official number
The death toll from the pandemic has been devastatingly high, with the official figure standing at 3.5 million people, but the World Health Organization estimates the real number could be up to three times higher. Here’s why.
Little-Known Illnesses Turning Up in Covid Long-Haulers
A significant number of post-covid patients suffer from syndromes that few doctors understand.
How well your immune system works can depend on the time of day
Our immune system is controlled by our "body clock" – an intricate 24-hour system which controls how cells function.
Nigerians turn to social media for encouragement
Social media has been much-maligned as a source of misinformation when it comes to vaccines. But for millions of Nigerians it is having the opposite effect, encouraging them to get their jabs.
World leaders and private sector commit to protecting the vulnerable with COVID-19 vaccines
In the COVAX Advance Market Commitment Summit, hosted by Japan, countries and industry came together to pledge support to the mission of making vaccines available equitably worldwide.
Change to cold-storage conditions could make Pfizer COVID-19 vaccine more widely available
Several regulators have agreed the mRNA vaccine can be kept refrigerated for up to 30 days, making it easier to distribute in lower-resource settings.
Keeping vaccines cool with cold chain
With millions of COVID-19 vaccines now being delivered around the world, they are utilising a huge hidden infrastructure, built up over decades and spreading across the globe, with one ultimate goal: keeping the vaccines cool. How did these…
What steps must be taken to secure oxygen - for COVID-19 patients and into the future
New waves of the COVID-19 pandemic in countries, such as Kenya and India, have exposed the poor management of oxygen supplies. Moina Spooner, from The Conversation Africa, asked Professor Trevor Duke, an expert on [oxygen provision] and editor of…
Malawi takes on religious beliefs that discourage immunisation
Malawi’s Ministry of Health is using its structures to reach out to a small section of religious groups that are threatening the success of the country’s immunisation programme.
Yellow Fever and the Panama Canal
At the turn of the 20th Century, US researchers in Cuba made the historic discovery that mosquitoes spread yellow fever. The finding was not only a medical breakthrough; it would also make possible one of the world’s greatest feats of engineering…
Could a new coronavirus be spreading from dogs?
Scientists think they may have caught a canine coronavirus in the process of adapting to humans.
Coronavirus: so many variants, but vaccines are still effective
Variants of interest and variants of concern seem to be popping up at an alarming rate. But how many of them do we really need to worry about?
How Yellow Fever decimated the USA’s first capital
In 1793, with the United States of America less than 20 years old, a yellow fever epidemic decimates the capital city, Philadelphia, and shines a spotlight on stark racial and social inequalities.
Why Africa’s critically ill COVID-19 patients have the world’s highest death rates
Death rates on the continent are higher than Asia, Europe, North America or South America, in large part because of a lack of resources and underuse of resources that do exist.
COVID-19: how rising inequalities unfolded and why we cannot afford to ignore it
It's been argued that pandemics are the great leveller, but with COVID the opposite is true – and we can't afford to ignore it.
The community health workers getting people vaccinated in Uganda
As Community Health Workers, Harriet Nankwanzi and Yusuf Maganda have built personal relationships with people in their communities who have over the years relied on their services to access vital health information.
Poo and the pandemic: How sewage is helping us fight COVID-19
Many countries are beginning to use wastewater monitoring to track COVID-19 outbreaks, but such sewage surveillance has a long history.
“If we don’t go out there, nobody would”: Vaccinating during an insurgency in Nigeria
Health workers go above and beyond the call of duty to ensure that the children of Borno State, Nigeria, are vaccinated.
The impact of COVID-19 on Malawi’s routine immunisation programmes
The pandemic is having an adverse effect on Malawi’s continued drive to provide all children with routine vaccines.
COVID vaccines: some fully vaccinated people will still get infected – here’s why
Reports of fully vaccinated people getting infected with coronavirus shouldn't cause alarm.
How "camel riders" are helping the COVID-19 vaccine push in Kenya
With tourism hit hard by the pandemic, camel riders in Northern Kenya have repurposed their herds to help spread the word about COVID-19 vaccines, using traditional healers to gain trust in the community.
The World Isn’t Getting Vaccinated Fast Enough. Here Are 4 Ways to Fix That
In an unprecedented show of global solidarity, the world came together to back COVAX, a unique global solution aimed at making equitable access to COVID-19 vaccines possible.
COVAX Manufacturing Task Force to tackle vaccine supply challenges
A COVAX Manufacturing Task Force has been established to identify and resolve issues impeding equitable access to vaccines.
Vaccinator training on smartphone screens amid COVID-19: Another “new normal”?
App-assisted vaccinator training is an approach that shows promise, write the team who ran a Gavi-supported pilot to trial a new screen-based learning mechanism in India. What’s more, amid the pandemic, the flexibility of learning from home,…
Gavi’s Seth Berkley named one of the world’s greatest leaders by Fortune
Dr Berkley is recognised alongside other world-changing vaccine champions in this year’s list for their part in the fight against COVID-19.
As the Spanish flu of 1918-1920 tore through America, a San Francisco mayor bet on the potential of face masks to contain the spread. Then, like today, the demand to mask up met resistance that blended distrust, ideology and gut feeling.
Covering COVID-19 in Guyana’s ‘Deep South’
The indigenous people adopted measures that were more applicable to the challenges but also their reality. And I tried to illustrate that in my story.
Mounting evidence suggests COVID vaccines do reduce transmission. How does this work?
Vaccination is likely to substantially reduce virus transmission by reducing the pool of people who become infected, and reducing virus levels in people who get infected.
Working towards vaccine equity to leave no one behind
Country and community perspectives on realising the targets of the Immunisation Agenda 2030.
Next Steps for a People’s Vaccine
The Biden administration’s decision to stop opposing a proposed COVID-19 waiver of certain intellectual-property rights under World Trade Organization rules is a welcome move. But ending the pandemic also requires scaling up knowledge and…
COVID-19 vaccines bring relief to Ugandans living with HIV
The pandemic has been tough for the 1.4 million Ugandans living with HIV, but the arrival of the COVID-19 vaccine has brought some welcome relief.
Expediting COVID-19 vaccination could prevent millions of deaths and save billions of dollars
Increasing vaccination coverage by even 1% can save thousands of lives and millions of dollars in medical and economic costs.
The US adds it support to patent waivers for COVID-19 vaccines
As countries worldwide are suffering from COVID-19 vaccine shortages, sharing intellectual property and know-how, and easing trade bans on the export of raw materials, could ease supply bottlenecks.
Weather forecasts a ‘life-saving tool against meningitis’
Meningitis affects more than 30,000 people in Africa every year. Scientists use weather data to predict location and scale of impending cases. The prediction helps country-level health services to plan emergency responses.
COVID vaccines: why waiving patents won’t fix global shortage – scientist explains
Increasing skills and the availability of raw materials would be a bigger boost for vaccine production right now.
Oxford vaccine professor: rich countries have a moral duty to share their COVID-19 shots
We cannot look back in the future and know we could have done more.
Emerging lessons from Africa’s COVID-19 vaccine rollout
With 47 African countries now rolling out COVID-19 vaccines and over 17 million doses given on the continent, early insights from Africa’s largest-ever immunization drive offer hope, inspiration and early, yet vital lessons. Here, we shine a…
Why Indian hospitals are running out of medical oxygen and how to fix it
Investment in bedside oxygen production could make hospitals more resilient in the face of COVID-19.
There will be no recovery without equity
The impact of COVID-19 threatens to reverse hard-worn strides in global health. Without immediate investments, we will miss our chance to save lives and help countries recover from the pandemic and build the human capital needed for a resilient…
Although it is only natural and proper for governments to focus on vaccinating their citizens first, it is important not to lose sight of the big picture. The only way we can beat COVID-19 is to defeat it everywhere, and the best way to do that…
Why routine immunisation is vital for pandemic preparedness
Trying to control a pandemic can feel like being in a warzone, yet even as we fight COVID-19 we still need to keep one eye on the future and potential emerging pandemics. Dr Velislava Petrova, senior manager in vaccine policy and investment at…
The Pandemic Within the Pandemic
Globally, antibiotic use in hospitals has surged since the start of the COVID-19 pandemic, and over-the-counter sales, legal in many countries, have soared. This is further fueling the global crisis of antimicrobial resistance, as bacteria evolve…
Understanding vaccine hesitancy: Cha’s story
A new documentary tells a powerful tale of families overcoming every obstacle to protect their children from vaccine preventable disease.
Helping children thrive through soap and vaccines
In Uttar Pradesh, India, over 1,000 children die every day from preventable diseases. Gavi’s long-standing partnership with Unilever and Lifebuoy has helped to reach 2.5 million people with essential vaccines and soap, while also leveraging the…
COVID-19 in India: an unfolding humanitarian crisis
Stories of reaching herd immunity were certainly premature.
An Indian Queen’s “Vaccine Selfie” in Oils
Why a 19th century portrait of three southern Indian noblewomen is “one of the most important scientific pictures in the history of medicine in India.”
Malaria vaccine achieves 77% efficacy in seasonal setting in study of African children
The R21/Matrix-M vaccine is the first to show promise of hitting the World Health Organisation’s target of 75% efficacy against malaria.
Why is India’s COVID-19 pandemic skyrocketing?
When the pandemic started, India defied expectations by having a milder outbreak than had been predicted. Now, it is seeing a deadly second wave that is bringing the country’s health system to its knees – so what is behind this, and how much is…
A cousin of Ebola, Marburg can kill nine out of ten people it infects, and international travel has taken it from Africa to Europe twice in the past 40 years. The virus has just hit Equatorial Guinea for the first time ever – what does this mean…
Mysterious Ailment, Mysterious Relief: Vaccines Help Some Covid Long Haulers
Scientists who study the post-illness syndrome are taking a close look at patients’ reports of this unexpected benefit of the vaccine.
Ivermectin: why a potential COVID treatment isn’t recommended for use
The antiparasitic drug was thought to be a potential treatment for COVID-19, but there isn't sufficient evidence to recommend its use, despite widespread support online.
Are We Risking a Debt Pandemic?
The prospect of recovery from the COVID-19 crisis makes it all the more urgent to have a firm vision of how the burden of public debt can be reduced once the coronavirus has been vanquished. For this reason, every country must work on itself and…
Resourceful Optimism: Fighting COVID-19 in Afghanistan
The pandemic has strained most countries’ health systems. Afghanistan has faced more hurdles than most.
Scientists have spent nearly 100 years searching for a better way to give vaccines
We didn’t have to be stuck with needles.
Serious blood clots “more likely” with COVID-19 infection than vaccine
Study suggests the risk of a serious brain clot is up to ten times higher in people with COVID-19 infections, compared to those receiving a COVID-19 vaccine.
COVAX: A new standard for global innovation and partnership
COVAX has a critical role to play in ending the acute phase of the pandemic. The initiative could also be a model for how we address some of the most urgent development challenges of our time, from climate change to poverty reduction.
“A ray of sunshine”: COVAX vaccines reinvigorate health workers in Eswatini
The delivery of vaccines has gone a long way to alleviating the fears and pressure on healthcare workers in Eswatini.
An expert explains how to track coronavirus variants
New Covid-19 variants could potentially jeopardise a lot of the work that has been done so far to contain the pandemic. Sonia Gonçalves explains how genomic surveillance can help us track and contain them.
COVID-19: does exercising really reduce the risk?
New research suggests people who exercise for 150 minutes a week are half as likely to die from COVID-19 compared to those who are consistently inactive.
COVID-19 ‘led to rise in stillbirths, maternal deaths’
Maternal health in low- and middle-income countries hit hard by COVID-19. Global review analysed stillbirth, maternal death rates from 17 countries. Maternity care must be prioritised during global health crisis, says report
As long as there have been vaccines, a vocal minority of public voices have made it their mission to rile up their communities against vaccination. In Montréal, in 1885, those voices were successful – and the results were deadly.
“I can only run away from death for so long”: Vaccinating Elders in Eswatini
The delivery of COVID-19 vaccines to Eswatini through COVAX means the elderly now have the chance to protect themselves against the disease. VaccinesWork visits a vaccination session in Lobamba.
Will summer slow the spread of COVID-19? New research sheds light
The seasons can affect transmission of the virus – but this will likely be dwarfed by the impact of public health measures.
How to Stop the Poverty Pandemic
Experience shows that innovative and evidence-based approaches, when executed well, can dent poverty. With the COVID-19 pandemic threatening to reverse hard-won global gains, the need for policy-relevant research, and for scaling effective…
What does it take to deliver COVID-19 vaccines?
Ending the COVID-19 crisis hinges on all countries being able to vaccinate their people. A lot goes into getting ready.
The World’s First Vaccination Campaign
At the turn of the 19th century, news of a 1796 medical breakthrough called 'vaccination' started to spread around the world. A fascinating recent book, War Against Smallpox by Michael Bennett, traces the subsequent dissemination of the actual…
Getting from zero to 100% – ensuring every child has routine immunisation
New data shows that investing in getting that first vaccination to children can be a turning point in getting them fully immunised.
Lockdown mental fatigue rapidly reversed by social contact, study finds
The study found people bounce back from their mental sluggishness soon after emerging from isolation.
A year of #VaccinesWork in 10 articles answering your most frequently asked questions
To mark one year of #VaccinesWork, we look back at some of the articles that answer the most frequently asked questions about COVID-19 during an unprecedented year for global health.
What is the blood clotting disorder the AstraZeneca vaccine has been linked to?
The European Medicines Agency has concluded that there is a possible link between AstraZeneca’s COVID-19 vaccine and very rare blood clots. But what are these clots and how great is the risk?
How geospatial technology is helping Nigeria’s COVID-19 vaccine roll-out
In a huge country like Nigeria, ensuring the right people receive COVAX vaccines is not just a question of how, but where. Could geospatial technology, trialled during previous polio campaigns, make a difference?
Bacteria 'shuffle' their genetics around to develop antibiotic resistance on demand
A genetic trick called an integron plays an important role in helping bacteria do this.
Should I get a COVID-19 vaccine if I’ve already had COVID-19?
Natural infection with the coronavirus that causes COVID-19 may not lead to long-lasting immunity, so it is important to get the vaccine as well.
The age of modern vaccines: An Abridged History of Vaccines, Part 2
As the 19th century entered its final quarter, “vaccination” meant one thing: immunisation with cowpox against deadly smallpox. By the end of the 20th century, safe and effective vaccines existed against 26 killer diseases. In the second part of…
Can a COVID-19 vaccine give me COVID?
Currently approved vaccines incorporate viral proteins or the instructions for making them, but they do not contain any live virus.
Immunization and vaccination experts from around the world outlined the current state of the world’s vaccination campaigns while addressing the urgent needs and concerns regarding public trust in the efficacy of vaccines; access and challenges.…
The end of the pandemic is coming – just don't set a date for the party
History tells us that the end of pandemics are rarely – if ever – neat, uncomplicated, or even easy to date.
The next pandemic: yellow fever?
In the 19th century, yellow fever had taken hold of parts of Europe and the USA, especially the Deep South, killing thousands. Now mostly in Africa and South America, this mosquito-borne disease could spread at any moment, threatening public…
Arriving at the First Vaccine: An Abridged History of Vaccination, Part 1
As early as the 1500s, efforts to halt the spread of smallpox included risky procedures designed to trigger immunity. By the 1800s, these methods – collectively termed “variolation” – had been supplanted by the earliest example of a new, safer…
How does resistance to disinfectants happen? We're on the road to answering the question
The increased use of disinfectants could allow for the development of bacterial strains which are resistant to disinfectants.
Somalia rolls out vaccines for COVID-19
On 15 March 2020, Somalia received 300 000 doses of Oxford AstraZeneca vaccines from the COVAX Facility to protect frontline workers and elderly people with chronic health conditions from COVID-19. The World Health Organization (WHO) hears…
Relieved and hopeful: Reflections after my first COVAX vaccine
In March, Mukami received her first COVAX vaccine dose. As a woman in her 60s living in Kenya, she has priority access to COVAX vaccines, alongside other high-risk groups like health care workers and people with underlying diseases. Here she…
What does water mean to the world?
On 22 March 2021, the world marked World Water Day, with the theme for this year’s celebration being “valuing water.” What can the long history of water’s connection with health teach us about its true value to the world?
How Can Covid Vaccines Be Safe When They Were Developed So Fast?
Experts say there’s nothing new about the research underpinning the covid vaccines and that they were tested in more participants than many other approved vaccines.
We know hand dryers can circulate germs through the air. Why are they still used everywhere?
What side are you on, paper or dryer? In either case, here's the bottom line on what to do after using the toilet.
Patient Zero: Understanding how new coronavirus variants emerge
By understanding the circumstances that make viral evolution more likely, we stand a better chance of staying a step ahead.
The COVAX Humanitarian Buffer Explained
COVAX was designed to ensure the most vulnerable in every country get access to COVID-19 vaccines. But what about people in conflict zones or humanitarian settings that can’t be reached by government vaccination campaigns? Last week Gavi approved…
COVID-19 vaccines: assessing country readiness
The key insights from the assessments to date present a high-level snapshot of country readiness to deploy COVID-19 vaccines based on initial findings from ongoing assessments in 128 countries.
A year of #VaccinesWork in 10 articles looking at vaccine-preventable diseases other than COVID-19
To mark the first anniversary of #VaccinesWork, we look back at some of the most read articles on diseases other than COVID-19 during an unprecedented year for global health.
Going universal: The search for an all-in-one coronavirus vaccine
Instead of working to make vaccines against each unique coronavirus or variant, what if we could provide broad protection across the field through an all-in-one coronavirus shot?
We must better protect wildlife to prevent future pandemics
Wuhan investigation points to wildlife as likely source of COVID-19. Climate change and illegal trade are increasing risk of zoonotic disease transmission. Wildlife protection, surveillance of zoonosis are key to early detection of ‘spillover…
“We’re closer to getting some normalcy back in our lives”: COVAX gets underway in Ghana
Ghana became the first African country to receive vaccines through the COVAX initiative in February. Meet one of the nurses spearheading the vaccination effort.
The next pandemic: H5N1 and H7N9 influenza?
More than 100 years after the 1918 Spanish influenza pandemic, type A influenza virus not only poses one of the largest threats to the modern world, but the risk of spill-over of avian influenza from poultry to humans is growing.
What we learned from tracking every COVID policy in the world
For one year, 600 people tracked 20 types of coronavirus restriction in 186 countries – here's what they found out.
The growing threat of pandemics
Whilst COVID-19 caught the world off guard, the speed at which it spread and the extent of its impact on people’s lives came as a great shock to most. However, some were not surprised at all.
Keeping It Cool: A visit to Ghana’s national vaccine storage facility
Ghana became the first African country to receive COVAX doses in February, kicking off a mammoth logistical effort in the country to get these doses to the frontline workers that need them. #VaccinesWork spoke to the cold chain experts making it…
A year of #VaccinesWork in 10 articles from around the world
To mark the first anniversary of #VaccinesWork, we look back at some of the most-read articles on how countries have been navigating COVID-19 during an unprecedentedly challenging year for global health.
These results can help allay previous concerns in Europe about a lack of trial data for older people.
The next pandemic: Chikungunya?
The tiger mosquito that carries the chikungunya virus is now moving into new habitats in Europe and America thanks to climate change. This means the once tropical disease could soon become a global pandemic threat.
An Ounce of Pandemic Prevention
The global response to the coronavirus pandemic, for all its flaws, has yielded impressive results by ignoring traditional bureaucratic and sectoral silos. That should spur us to raise our ambitions for global public health, with an emphasis on…
What is Africa’s vaccine production capacity?
Accra, 18 March 2021 – COVID-19 vaccination in Africa is gathering pace, with more than 7 million doses so far administered. But the continent received vaccines later than other regions of the world and in limited quantity. A few weeks after…
“I serve my nation through my profession”: Community vaccinators in Afghanistan
Community vaccinators go where fixed health centres can’t reach. In Afghanistan, that means fighting Taliban injunctions, entrenched gender imbalances and difficult terrain, all to ensure the nation’s children are protected.
Tariah Adams: on tap for women and girls’ right to water, sanitation and hygiene
In today’s blog post, we’re featuring a fierce and passionate female WASH advocate from Nigeria. Tariah Adams, a Senior Communications and Advocacy Officer for White Ribbon Alliance Nigeria and campaign mobilizer, she has a personal interest in…
At the end of the year 2019, we created an elaborate 2020 plan for our projects at Beyond the Classroom Foundation. As we got closer to our second project scheduled for the end of March, the government imposed a lockdown, banning all public…
Ebola has so far only affected African countries, and occasional cases outside of the continent have been rapidly contained. But the virus could mutate to spread more easily between people, making it more of a pandemic threat.
A year of #VaccinesWork in 10 articles celebrating women in global health
To mark the first anniversary of #VaccinesWork, we look back at some of the most read articles on COVID-19 during an unprecedented year for global health.
So far, homeschooling has been trial and error because it’s difficult to keep the twins calm and engaged for long periods of time. We have no fixed curriculum and use online educational programs as a guide.
“We’ll all be fine”: COVID-19 vaccines arrive at Korle-Bu Teaching Hospital, Accra
After a difficult year, the vaccination roll-out is underway at Ghana’s premier medical facility, where health workers dare to hope that things can go back to normal.
John Nkengasong on Africa's Vaccine Game Plan
John Nkengasong is the Director of the Africa Centers for Disease Control and Prevention. He is also the WHO special envoy for Africa. John spoke to CovidHQAfrica desk about a range of issues including his vision for the vaccines in Africa, and…
No "us versus them": why equitable inclusion of all migrants in COVID-19 vaccine plans is essential
COVID-19 has demonstrated that viruses not only know no borders, but they also do not discriminate based on immigration status. Failing to take migrants into account in our vaccination efforts would hamper the effectiveness of these campaigns and…
A year of #VaccinesWork in 10 COVID-19 articles
To mark the first anniversary of #VaccinesWork, we look back at some of the most read articles on COVID-19 during an unprecedented year for global health.
Should I invest in a pulse oximeter?
Sales of medical devices that measure the oxygen saturation of blood are booming thanks to the COVID-19 pandemic. But are they worth the investment?
Dormant virus reawakening may have triggered Ebola outbreak
Genetic sequencing has linked the recent outbreak of Ebola disease to a survivor of the 2014-16 West Africa epidemic, meaning the virus could have laid dormant in one survivor for over five years. What does this mean for our efforts to control…
Nigeria rallies over 7,000 traditional leaders against COVID-19
Abuja, 12 March 2021 – As the second wave of COVID-19 affects the global health and economic community, the World Health Organization (WHO) is supporting government to engage strategic stakeholders as part of concerted efforts to reverse the…
How can supply keep up with demand for COVID-19 vaccines?
With the historic global roll-out of COVID-19 vaccines in full swing, bottlenecks in the supply chain are inevitable as manufacturing output tries to keep pace with demand. A manufacturing supply chain summit last week outlined potential…
Vaccine Altruists Find Appointments for Those Who Can’t
An army of volunteers help people who otherwise would have had difficulty securing a covid vaccination because of cumbersome computer or telephone registration systems.
Battling misinformation wars in Africa: applying lessons from GMOs to COVID-19
For anyone who has worked on crop improvement in Africa over the last three decades, the flood of misinformation around vaccines evokes an eerie sense of déjà vu.
The next pandemic: Nipah virus?
Nipah virus can kill as many as three out of four people it infects. Since the fruit bat that carries the virus often comes into contact with humans, here we explain why the development of drugs and vaccines for the disease is becoming…
'Like being hit with a cricket bat’: A doctor’s battle with long COVID
Dr Paul Garner, a British infectious disease expert at the UK’s Liverpool School of Tropical Medicine caught COVID-19 early in 2020, and then developed Long COVID. Here, he talks frankly with Gavi about his experiences, how he recovered and how…
When can children get the COVID-19 vaccine? 5 questions parents are asking
COVID-19 vaccine testing on children is just getting started, and only in adolescents.
Why Ebola is back in Guinea and why the response must be different this time
The virus is always present in nature and when circumstances allow, it may jump from one species to another.
Since COVID-19 was declared a global pandemic, many countries around the world imposed some form of quarantine to control its spread. What can the history of quarantine teach us about isolation and lockdowns now?
Diphtheria may resurface as a ‘major global threat’ study warns.
Researchers identify bacterial variants that may be evolving resistance to antimicrobials and vaccines.
Ten lessons from Ogun State - from first COVID-19 case in Nigeria to building a resilient response
Vivianne Ihekweazu (Lead writer): On the day marking one year since the first COVID-19 case was reported in Nigeria, this article reflects on the experience and lessons learnt from the Honourable Commissioner for Health, Dr. Tomi Coker as she led…
Don’t overlook the most important metric for Johnson & Johnson’s Covid-19 vaccine
Overall efficacy is just one metric.
AstraZeneca vaccine: careless talk has dented confidence and uptake in Europe
Stockpiles of this vaccine are going unused in France and Germany, and unfounded criticism of it may be partly to blame.
Keeping the Cold Chain Cold: The Importance of Maintenance
This is the third in a three-part series highlighting key lessons learned since the launch, four years ago, of the Cold Chain Equipment Optimization Platform (CCEOP), established by Gavi, the Vaccine Alliance, to ensure optimal cold chain…
Private sector innovation meets public health expectations
This is the second in a three-part series highlighting key lessons learned since the launch, four years ago, of the Cold Chain Equipment Optimization Platform (CCEOP), established by Gavi, the Vaccine Alliance, to ensure optimal cold chain…
Breaking the glass vial: Women at the forefront of COVID-19 vaccine
Leaders in global health have been working on COVAX – a global initiative to ensure the fair distribution of COVID-19 vaccines. Some of the top jobs are held by a diverse group of women, so in honour of International Women's Day we'd like to…
International Women's Day 2021
For this year's International Women's Day VaccinesWork has interviewed a number of women leaders and front line workers who represent this year's IWD theme: #ChooseToChallenge
International Women’s Day 2021: “Women come to me and I guide them”
This International Women’s Day Vaccines Work is hosting a series of interviews with inspirational women from across the world. Here Nighat Rani, a vaccinator in Pakistan, shows how her work rests on a bedrock of female empathy and understanding…
International Women’s Day: Chizoba Wonodi on the importance of a good mentor
This International Women’s Day Vaccines Work is hosting a series of interviews with inspirational women from across the world. Here Professor Chizoba Wonodi, Founder of Women Advocates for Vaccine Access in Nigeria and Country Director at the…
International Women’s Day: Quarraisha Abdool Karim on the fight against HIV
This International Women’s Day Vaccines Work is hosting a series of interviews with inspirational women from across the world. Here Professor Quarraisha Abdool Karim, epidemiologist and associate scientific director for CAPRISA – the Centre for…
International Women’s Day: Heidi Larson and the fight against vaccine hesitancy
This International Women’s Day Vaccines Work is hosting a series of interviews with inspirational women from across the world. Here Professor Heidi Larson, Founding Director of the Vaccine Confidence Project, explains how gender can affect…
International Women’s Day: Caring for Everyone: an ASHA worker’s COVID-19 story
This International Women’s Day Vaccines Work is hosting a series of interviews with inspirational women from across the world. Here Rathnamma P, an 'ASHA' in Bengaluru, India, explains how the country's army of one million female community health…
International Women’s Day: Anuradha Gupta on ‘choose to challenge’
This International Women’s Day Vaccines Work is hosting a series of interviews with inspirational women from across the world. Here Anuradha Gupta, Gavi’s Deputy CEO, explains what ‘choose to challenge’ – this year’s IWD theme – means to her.
The value of COVID-19 vaccines in children: Roadmap for a safer world
To make the world safe from COVID-19, we need everyone vaccinated, including children. To get back to normality, we also need vaccinated adults, ongoing safety measures, safe re-opening of schools, access to all recommended vaccines – and…
A little less hesitation, a little more action - Elvis and the polio vaccine
As political and public health leaders across the world work to encourage people to get vaccinated against COVID-19, what can they learn from the King of Rock ‘n’ Roll getting jabbed?
The ins and outs of Kenya's COVID-19 vaccine rollout plan
Reaching the goals of the plan requires the best possible interaction between public and private -for profit and not-for-profit - healthcare sectors.
Drone delivered COVID-19 vaccines take to the air
The first deliveries of up to 2.5 million doses of COVAX vaccines by autonomous drone began this week in Ghana.
What is COVID-19 vaccine efficacy?
Several vaccines that have become available have shown different levels of efficacy. So what is vaccine efficacy? Dr Lee Hampton, a paediatrician and medical epidemiologist with Gavi, the Vaccine Alliance, explains.
Scientists edge closer to a universal flu vaccine
A vaccine that apparently protects animals against multiple strains of flu has been created by tethering a relatively unchanging part of the surface proteins found on the virus to nanoparticles.
How to make sure COVID-19 vaccines reach as many people as possible
As more COVID-19 vaccines become available, countries the world over are now faced with the daunting task of carrying out mass vaccinations. Here, one expert explains how her organisation is supporting rollouts in the US and offers insights…
COVID vaccines: how to make sense of reports on their effectiveness
Real-world studies of vaccines aren't directly comparable with clinical trials, but their results are still good news.
The first COVAX vaccinations begin
COVID-19 vaccination campaigns using COVAX-funded doses commenced today in Côte d’Ivoire and Ghana, the first global rollout.
Country readiness for COVID-19 vaccines
This article is part of a series of explainers on vaccine development and distribution. Learn more about vaccines – from how they work and how they’re made to ensuring safety and equitable access – in WHO’s Vaccines Explained series.
COVAX vaccines take to the air by drone
Ghana is not only the first country to receive a shipment of COVAX vaccines, but it will be the first to broaden its reach by delivering them by drone. The new partnership with Zipline and UPS Foundation will see 2.5 million doses delivered this…
COVID-19: what happens if some countries don't vaccinate?
Even those that live in areas where the population has already been vaccinated would not be totally protected if the virus mutates elsewhere.
Planning for Success: Project Management Teams for the CCEOP
The first in a three-part series of blogs highlighting key lessons learned since the launch, four years ago, of the Cold Chain Equipment Optimization Platform (CCEOP), established by Gavi, the Vaccine Alliance, to ensure optimal cold chain…
How Roald Dahl became a passionate vaccine advocate
Roald Dahl's daughter Olivia died of measles aged seven.
Why vaccine side effects might be more common in people who've already had COVID-19
Minor side effects are a normal sign that the immune system is mounting a protective response following vaccination, although they aren’t universal.
Ebola strikes West Africa again: key questions and lessons from the past
Countries in the West Africa region are in a very different position to seven years ago. They now have the experience of the past as well as new tools to tackle Ebola.
Real-world data supports the use of AstraZeneca vaccine in older individuals
Vaccine data from Scotland provides reassurance that the Oxford-AstraZeneca and Pfizer-BioNTech vaccines will significantly reduce hospitalisations and deaths from COVID-19 among older people after the first dose.
African countries have an advantage in rolling out Covid-19 vaccines
The continent is one of the most experienced regions in the world in dealing with disease outbreaks.
Why do antibodies fade after a COVID-19 infection, and will the same thing happen with vaccines?
Maintaining antibodies in the blood requires creating certain long-lasting immune cells – but this doesn't always happen.
Will we ever get rid of COVID-19?
The pandemic won’t last forever, but the virus that causes COVID-19 might. Here we look at what we can expect over the next couple of years in a Q&A with Dr Lee Hampton, pediatrician and medical epidemiologist at Gavi.
How long does immunity last against Ebola?
The tracking of antibodies in Ebola survivors suggests they wax and wane, which could make vaccine booster shots necessary.
Saving the planet from catastrophic climate change will require not only a dramatic increase in funding for clean-energy research and development. We need innovation in policy just as much as in technology.
The $4 trillion economic cost of not vaccinating the entire world
The world's most advanced economies will incur half the total costs associated with a failure to vaccinate poorer nations, which could exceed $4 trillion if only half their citizens are inoculated.
Tackling pandemic threats proactively
If we are to minimize the impact of future disease outbreaks, we must recognize the importance of immunization and invest in preemptive vaccine development, writes Tim Keys.
100 years and counting of mask wearing in Japan
As wearing face masks in public becomes the new normal across the world, what can we learn from a country like Japan, where this has been a long-established practice?
Grab, jab and release: keeping rabies off the streets of Goa
The global effort to contain COVID-19 risks disrupting campaigns against other diseases. Will the fight to control the deadly rabies virus in India be among them?
Vaccines against SARS-CoV-2 will have side effects – that's a good thing
The side effects of new SARS-CoV-2 vaccines are a result of immune system activation. While uncomfortable, they are both normal and expected. They are a sign that the vaccine is working.
The G7 has an opportunity to demonstrate global leadership in the COVID-19 pandemic by making the success of the international COVAX vaccine-access facility its top priority. Global solidarity is not only morally right, but also offers the…
How have Covid-19 vaccines been made quickly and safely?
Unprecedented international cooperation and focus have led to multiple effective and safe Covid-19 vaccines in less than a year, and created a blueprint for future vaccine development. Here's how
Why having COVID-19 data on men and women is critical
COVID-19 deaths differ among men and women but the differences are not reported. A project is building sex-aggregated data on COVID-19 globally. Such data is key to developing effective solutions to fight COVID-19.
Side Effects and COVID-19 Vaccines: What to Expect
It’s totally normal to experience side effects from COVID-19 vaccines. Here’s what you need to know.
Could new outbreaks put Ebola vaccines to the test once again
Guinea is having its first Ebola outbreak since 2016, when West Africa experienced the biggest outbreak ever seen. Could the vaccine that contained previous outbreaks soon be redeployed?
When will the world be vaccinated against Covid-19?
With the first Covid-19 vaccines in short supply, how do governments decide who gets a vaccine first?
Is the colonial era still impacting people’s health today?
A new study suggests that historical traumas from the French colonial era may be associated with less trust in modern medicine and lower vaccination rates today.
COVID-19 variants are not going away, but vaccines may help make that OK
Clinically significant SARS-CoV-2 variants are likely to be here to stay, but multiple COVID-19 vaccines still have a role to play.
All roads lead to Ocean: how cancer treatment works in Tanzania
In Tanzania, the Ocean Road Cancer Institute is doing its part to ensure that people across the country can receive cancer treatment and care, regardless of their ability to pay.
Maina Modu is the programme manager for Routine Immunisation in Borno State, working with the State Primary Health Care Development Agency (SPHCDA). He coordinates the state emergency routine immunisation coordination centre activities.
Tackling the COVID Hunger Crisis
The choice facing world leaders is simple: act now to tackle the hunger crisis, or pay a much higher price later. Immediate action will be cheaper and save more lives than responding only after multiple famines have taken hold and a generation’s…
Community Health Workers, Often Overlooked, Bring Trust to the Pandemic Fight
As the pandemic brings long-standing health disparities into sharper view, community health workers are coming to the forefront in the public health response. This fast-growing workforce help fill the gaps between health care providers and low-…
A leap forward in vaccine technology
By building on the lessons learned in 2020, it should be possible in the long term to compress vaccine development timelines still further. That would allow healthcare systems to stamp out disease outbreaks much earlier and save many more lives…
The Silent Pandemic of Antibiotic Resistance
Antibiotic resistance has been a slow-growing scourge, fueled in part by relatively weak political support for implementing national action plans and surveillance systems. Because the problem is essentially the result of multiple systems failures…
All aboard the vaccine minibus: getting immunisation back on track in Pakistan
In Pakistan, a life-saving game of immunisation catch-up is underway after the COVID-19 emergency forced over a million children to miss out on routine vaccinations.
How important are surfaces in the transmission of COVID-19?
Contaminated doorknobs and packaging probably aren’t the main way the coronavirus spreads. But don’t throw away the hand gel and cleaning products just yet.
Will coronavirus really evolve to become less deadly?
The coronavirus is evolving, but which path it will take is far from certain.
The world's last smallpox patient
Smallpox was one of the deadliest diseases in the world, but it’s the only one in history to have been eradicated globally. One Somali man sat on the frontline of this effort and can inspire the world today as we battle yet another devastating…
What’s the most effective face mask for preventing COVID-19 transmission?
Face coverings have become a standard feature of pandemic life, but which mask is best for everyday use, and how should you take care of it?
Protecting Lower-Income Countries with COVID-19 Vaccines Requires Global Solidarity
The medical and moral imperative for equitable access to COVID-19 vaccines is why COVAX was created. Co-led by Gavi, the Vaccine Alliance, together with the World Health Organization (WHO) and the Coalition for Epidemic Preparedness…
4 things about mRNA COVID vaccines researchers still want to find out
Researchers are already working to improve the current crop of mRNA vaccines. Hopefully this will help them become more practical and affordable for the entire world, not just first-world countries.
How the Oxford-AstraZeneca vaccine could still help the battle against the COVID-19 variant
Research suggesting that one of the COVID-19 vaccines provides only minimal protection against mild-moderate infection caused by the 501.Y.V2 variant has raised concerns. But this doesn’t mean it won’t prevent severe disease and deaths.
Eliminating cervical cancer depends on global effort to ensure supply meets demand
Global access to HPV vaccine is vital, particularly in lower-income countries. Lessons learned from HPV roll-out could boost uptake of COVID-19 vaccines.
The Oxford-AstraZeneca COVID-19 vaccine could stop transmission of the virus
Several COVID-19 vaccines may have now demonstrated their ability to prevent disease, but it was still not clear whether any could stop the virus being passed on. Now however a preliminary study suggests that some might also be able to reduce…
Can the world ever be cervical cancer-free?
As World Cancer Day approaches amidst the COVID-19 pandemic, it’s now more important than ever to ensure continued access to HPV vaccines to eliminate cervical cancer around the world.
Women Leaders in Polio Eradication: Dr. Alda Morais Pedro De Sousa
Dr. De Sousa has spent more than twenty years charting the highs and lows of polio eradication in Angola.
Everything we know about Johnson & Johnson’s single-dose Covid-19 vaccine
The single-dose vaccine showed promising results in preliminary data from a phase 3 clinical trial.
The next step: Lifesaving typhoid conjugate vaccines reach Punjab province, Pakistan
Punjab province introduces typhoid conjugate vaccine, taking the next step in protecting children from typhoid in Pakistan.
Results from Novavax vaccine trials in the UK and South Africa differ: why, and does it matter?
The results indicate that the vaccine efficacy in the UK was 89% for individuals who received at least two doses of vaccine. In South Africa, the vaccine efficacy was 60% in people without HIV.
With the World Health Organization inviting COVID-19 vaccine manufacturers to submit their candidates for evaluation, we examine the process from submission to Emergency Use Listing.
COVID-19 variant found in Brazil ‘spreads faster’
Manaus variant spreads faster and could carry higher risk of infection. Researchers believe new variant spread from Brazil to Asia. Fast, widespread vaccination is best way to slow down mutations.
Q&A: Tales from the COVID frontline – coping with the pandemic on a psychiatric ward
Irene Baker* is a care assistant at a psychiatric hospital in England. She describes the challenges of caring for mentally ill patients during the second wave of the COVID-19 pandemic.
The body's fight against COVID-19 explained using 3D-printed models
A biologist explains what proteins do in viruses, how they interact with human cells, how the vaccine delivers mRNA into the cell and how antibodies protect us.
New GPEI Director Aidan O’Leary takes helm of global polio effort
In a special one-off interview, PolioNews (PN) talks to both Aidan O’Leary (A-O’L) and Michel Zaffran (MZ) about the future of polio eradication.
Pfizer and Moderna’s COVID-19 vaccine candidates have turned the vaccine cold chain into hot news. But what is a cold chain? How does it work? And what does it have to do with equity?
4 of our greatest achievements in vaccine science (that led to COVID vaccines)
We've gone from a novel virus to several COVID-19 vaccines in less than a year. Here's what we've learned from earlier vaccines to allow this to happen.
COVAX Supply Forecast reveals where and when COVID-19 vaccines will be delivered
COVAX is forecasting it will roll out over 2 billion COVID-19 vaccine doses in 2021. What does that mean for the world’s poorest countries?
How much of an impact do vaccines really have?
The world’s most comprehensive study reveals all! A new study looking at the impact of vaccinations in 98 countries over the past 20 years concludes that 37 million people are alive today thanks to vaccines.
Will the Covid-19 vaccine work on the new variants?
Scientists are working to understand what mutations of the virus mean for inoculation strategies.
How will COVID-19 vaccines be approved for use in Australia?
Australia is set to get the green light to roll out the Pfizer vaccine any day now. There is a complex process behind this.
The state of the world’s sanitation
To achieve universal sanitation, we need greater investment and higher rates of sanitation coverage.
Is there an economic case for global vaccinations?
New research adds to the argument that the global distribution of COVID-19 vaccines is the most optimal solution for a global economic recovery.
What if I don’t get my second Covid-19 vaccine dose on time?
Don't panic if a second dose isn't available exactly when you need it.
In Zimbabwe, Friendship Bench, a community-based approach is providing solutions to people struggling with their mental health during this pandemic.
The importance of blood markers in assessing vaccine efficacy
If vaccine efficacy can’t be assessed through placebo-controlled trials in future, how can we know if they are effective?
Hesitancy over new COVID-19 vaccines doesn’t seem likely to affect uptake
The initial concern that nervousness over the new vaccines would slow uptake doesn’t seem to have become a reality in the early days of COVID-19 immunisation, although it will be critical to maintain vaccine confidence suggests an Ipsos poll.
South African scientists who discovered new COVID-19 variant share what they know
Scientists have observed that 501Y.V2 has quickly become "dominant" among multiple variants that have been circulating in the South African population.
Door to Door in Miami’s Little Havana to Build Trust in Testing, Vaccination
It’s time-consuming but worthwhile: Residents respond to messages about Covid testing and vaccines when outreach teams speak their language and make a personal connection.
COVID-19 and the cost of vaccine nationalism
Without a vaccine, the worldwide economic impact of COVID-19 would have been $3.4 trillion a year. But even with a COVID-19 vaccine, unequal allocation could cost the global economy up to $1.2 trillion a year in GDP.
Why healthy food and its local production should be part of the COVID-19 response
COVID-19 is deepening global food insecurity, as the pandemic’s economic impact adds to existing challenges.
Vaccines that use harmless viruses as a delivery mechanism are vulnerable to being attacked by our immune system – but experimenting with how they are given could get around this.
Delaying the second COVID vaccine dose – a medical expert answers key questions
A medical professor explains the reasoning behind the delay in the UK and what impact this might have on the vaccine's effectiveness.
Could coronavirus persist in ‘safe havens’ of the body?
Some people continue to shed viral RNA for weeks or months after developing COVID-19. Could persistent pockets of infection be to blame?
The public health impact of a single-dose HPV vaccination schedule
New review of evidence on a single-dose HPV vaccination schedule is published by PATH-led consortium.
COVID-19 policy briefs must be realistic: a review by young southern African scientists
African leaders can make strategies to fight COVID-19 more accessible to the people.
What it takes to vaccinate 39 million children in Pakistan
Health workers take COVID-19 precautions to deliver polio drops.
The Ethics of Prioritizing COVID-19 Vaccination
In the United States and some other countries, members of disadvantaged racial and ethnic minorities have a lower-than-average life expectancy, and therefore are under-represented among those most likely to die from COVID-19. How should…
Breaking down barriers: UNICEF volunteers lead sanitation survey across Bangladesh
Young volunteers gain new skills and give back to their communities.
Mutating coronavirus: reaching herd immunity just got harder, but there is still hope
New variants will push the number needed to reach herd immunity up.
South Sudan: We are ready to take up the challenge
Western Equatoria Ministry of Health and UNICEF are preparing for COVID-19 vaccination campaigns.
Eureka! Two Vaccines Work — But What About the Also-Rans in the Pharma Arms Race?
How two effective vaccines on the market make it so much harder to quickly test any competing vaccines.
How COVID-19 is placing increased pressure on water resources for farmers in Cameroon
In Cameroon’s Marua region, the pandemic means farmers are now faced with the double challenge of using what limited water resources they have to grow crops while saving lives through handwashing.
Oxford scientists: how we developed our COVID-19 vaccine in record time
What normally takes decades has been achieved in 12 months, without cutting corners.
Has Covid-19 permanently altered the development timetable for other vaccines?
Covid-19 has changed the world of vaccines, but that doesn't mean all diseases will get treatments as quickly.
Preparing ahead: How Imo State harnessed the REDISSE project to improve its COVID-19 response
As at January 12, 2021, Nigeria has had over a hundred thousand confirmed cases of the novel 2019 coronavirus disease (COVID-19) infection. The pandemic has continued to challenge healthcare system...
As long as humans encroach on nature, pandemics are inevitable — making it important to concentrate resources in areas where people and wildlife are linked.
What is it like to receive two doses of a COVID-19 vaccine? One 92-year-old shares his experience.
On 8 January, Derrick C became one of the first people in the UK to receive his second dose of a COVID-19 vaccine, three weeks after the first dose. Here we ask him what that was like.
How to keep Ebola vaccines at -70°C?
As a Gavi-funded emergency stockpile of Ebola vaccines becomes accessible, we look at the ultra-cold chain equipment used to keep these vaccines at the right temperature.
Can you spread Covid-19 if you get the vaccine?
Answering this question will take us one step closer to our new normal.
Why resistance is common in antibiotics, but rare in vaccines
How resistance to drugs originates, and why it's different for vaccines.
COVID-19 immunity: how long does it last?
Long-term protection will depend on the 'memory response' developed by our immune systems – and the initial signs are promising.
Lessons from Rwanda’s Fight Against COVID-19
While some of the world's richest and most technologically advanced countries have struggled to contain the spread and morbidity of COVID-19, Rwanda has set a shining example of how to manage a pandemic and safeguard public health. Chief among…
Why delivering COVID-19 vaccines might be just as hard as developing them
From Liability Laws to Production Delays, the 2009 H1N1 Swine Flu Vaccine Rollout Offers a Cautionary Tale for Today.
Coronavirus: few vaccines prevent infection – here’s why that’s not a problem
Sterilising immunity means that the immune system is able to completely prevent a virus from replicating in your body. Not all vaccines provide this.
Trekking through the snow to deliver vaccines
Vaccinators tackle winter conditions and challenging contexts during Afghanistan’s last polio campaign of the year.
COVID-19 comes as a double blow to those living with HIV
In March 2020, as soon as the first cases of COVID-19 were detected in Kyrgyzstan, a state of emergency was declared, and the country went into lockdown.
Can Poor Countries Avoid a Vaccine Bidding War?
For all of the good news about the arrival of safe and effective COVID-19 vaccines, the hard truth for the coming year is that global demand will outpace supply. Without a multilateral agreement to allocate doses globally, the road to recovery…
Is altering the dosing strategy of COVID-19 vaccines a good idea?
Uncertainty in the availability of vaccine doses is prompting some countries to consider altering dosing schedules, or mixing different vaccine types. How could this impact the effectiveness of vaccines?
COVID-19 FAQs: The 10 questions you need to know the answer to
We believe the best way to address misconceptions is to arm yourself with the facts and have a few key points and statistics. Here’s our list of facts to combat common misinformation.
Coronavirus vaccines: how will we know when life can go back to normal?
Here's what we still need to find out before we can know when we'll be able to return to our pre-coronavirus ways.
In less than a year, the world has come together to develop effective COVID-19 vaccines and a multilateral platform for allocating them most efficiently around the world. But with the risk of vaccine nationalism still looming large, now is the…
Long-lasting symptoms appear unrelated to how bad your infection was, though women appear more affected than men.
Managing Well in the Work-From-Home Era
Managers owe it to their employees to stop treating work from home like a luxury. The office wasn’t invited into the home. It turned up like an unexpected guest – and it shows few signs of leaving soon.
How close are we to a ‘workable’ HIV vaccine?
Despite huge advancements in the treatment and prevention of HIV/AIDS in recent decades, an effective vaccine remains elusive – and is desperately needed to end the global pandemic that kills more than 700,000 people each year.
Severe COVID may be caused by 'autoantibodies' – here is what that means
Antibodies that go rogue and attack healthy tissue identified in patients with severe COVID.
Rethinking healthcare in Africa with geospatial mapping
In August 2020, Nigeria became the last African country to be declared free from wild poliovirus.
Five mantras for effective COVID-19 vaccine communication
Well-designed communication can increase healthy behaviours, including vaccine uptake. Here are our top five mantras for how to think about COVID-19 vaccine communication.
MVIP update – 1 million doses administered, Kenya 1st anniversary, cooperation for vaccine access
Kenya marked its 1st anniversary of the launch of the pilot in September, with more than 128,000 children reached with vaccine, and one country health official expressing “a great sense of pride” in being part of the effort to protect children…
A united front: building vaccine confidence during a pandemic
To combat vaccine misinformation at home and around the globe, we must build trust.
To be effective, COVID-19 vaccination plans must include migrants
As we prepare for one of the world’s largest vaccination efforts, and on the occasion of International Migrants Day, we are coming to a critical reckoning: the need for inclusive approaches in our health-related thinking and practices has never…
Ask an Expert: Why Are There So Many COVID-19 Vaccines — and Is It Better to Have More?
Vaccines have been approved – so why are we still developing others?
Why get vaccinated when the flu vaccine doesn’t work well?
The best way we can prevent flu infection is by getting vaccinated. The problem with the flu vaccine is that in some years it doesn’t work as well as others.
More typhoid conjugate vaccines, more impact
A second typhoid conjugate vaccine has achieved WHO prequalification and others are in development. More vaccines will help increase access to TCVs.
2020 Year in Review: The impact of COVID-19 in 12 charts
12 charts provide an overview of our research in the face of a truly unprecedented crisis.
Will an mRNA vaccine alter my DNA?
Some of the COVID-19 vaccines use messenger RNA to provoke an immune response. But what exactly is this genetic material, and how does it interact with the DNA in our cells?
A 4-point checklist for assessing countries' vaccine readiness
As the new COVID-19 vaccines begin to ship out, how ready are health systems to manage delivery? Here are four ways countries should frame that question.
Getting girls on board with the HPV vaccine
New girl-centered toolkit helps health officials develop and implement communications campaigns that resonate with girls in their countries.
Rallying to vaccinate every child against polio
Community mobilizers inform and prepare communities for vaccinators.
What psychology can tell us about why some people don’t wear masks – and how to change their minds
Some people respond strongly to perceived threats to their freedom and push back – others are simply more accepting of risk.
Are African countries ready for the COVID-19 vaccine?
Preparing African countries for COVID-19 vaccines will require thoughtful planning and unprecedented coordination across a wide-range of stakeholders.
We need more data on the low-dose, high-dose regimen used in one arm of the trial, which may make the vaccine more effective.
Mobile phone data reveals the most effective strategies for reducing COVID spread
Restaurants, gyms and religious establishments appear to account for most coronavirus infections in US cities, but new analysis suggests there are strategies which can reduce the risks.
Could Ebola survivors hold the key to understanding long COVID?
Like patients with long COVID, some Ebola survivors have lingering symptoms which can make it difficult to work or function in everyday life. Dr Janet Scott has been studying these survivors, and now also has her sights on COVID-19. She tells us…
What if I’ve already had COVID – do I still need a vaccine?
Routine vaccinations during a pandemic – benefit or risk?
Some countries may stop their vaccination programs for a while to reduce the risk of spreading COVID-19. But which is better: fewer coronavirus infections or making sure children get all their usual vaccinations?
Without a greater focus on the gender dynamics of the COVID-19 crisis, women and girls will suffer long-term handicaps that constrain their economic prospects for years to come, if not permanently. We already know enough about the pandemic's…
Keeping trust in immunisation during the COVID-19 pandemic
At this crucial moment for immunisation, considering community perceptions of vaccination has never been more important.
Health workers use special days to reach every child with life-saving vaccines
Not even the COVID-19 pandemic will stop them.
“Don’t use COVID-19 as an excuse not to immunize your children, you will regret it.”
Janet, mother of twins, tells fellow mothers.
Five ways that scientists are ensuring the safety of COVID-19 vaccines
As companies race to develop COVID-19 vaccines, some development processes have been run in parallel to stop the pandemic as quickly as possible. Yet safety remains paramount; now that we are on the brink of rolling out some of the vaccines that…
Why an antidepressant could be used to treat COVID-19
Fluvoxamine has shown positive results in early trials.
The peril of ignoring childhood pneumonia in the age of COVID-19
The COVID-19 pandemic now threatens global progress on preventing and treating childhood pneumonia, potentially reversing decades of health gains for the world’s most vulnerable children.
Four reasons why we need multiple vaccines for Covid-19
Having a range of Covid-19 vaccines available for people to use around the world will be essential to bringing the pandemic under control. Here’s why.
10 things you should know about vaccine candidates
Despite the benefit of all the recommended public health measures in preventing transmission, vaccines still provide us with the best chance of our lives returning to some semblance of normality.
Equal, rapid access to COVID-19 vaccines won’t just save lives; it will save money
Research suggests that the quickest way to end the pandemic and limit the economic damage is for all countries to benefit from a coordinated global vaccine deal.
IOM and Gavi working together to give the most vulnerable ‘a good start’ in South Sudan
This week Gavi signed a historic agreement with the International Organization for Migration (IOM) to improve immunisation coverage for migrants and forcibly displaced persons around the world. IOM’s South Sudan Communications Officer, Liatile…
Why the Oxford AstraZeneca vaccine is now a global game changer
The global pandemic is far from over, but this latest in a series of welcome announcements brings renewed hope even if several hurdles remain.
The longest mile in the COVID-19 vaccine cold chain
Promises of COVID-19 vaccine doses for developing countries will face challenges along the crucial cold chain.
Oxford vaccine results are in: here's how to ensure it is used
The Oxford vaccine – developed in partnership with AstraZeneca – stops 70% of people developing COVID symptoms, and, depending on how the doses are given, may even protect up to 90% of people.
Resumed immunization campaigns are critical to preventing COVID-19 as well as polio
Local adaptation and innovation has allowed the resumption of polio immunization campaigns – and points the way to more effective, community-based delivery of a whole range of basic health services.
COVID-19 antibody levels could fall faster in men than women
New research suggesting that antibodies reduce faster in men compared with women could have implications for vaccine development.
Although antimicrobial resistance has been a known and growing problem for decades, only one new class of antibiotics has been discovered since 1984. Tackling AMR requires a fundamental change in how new antibiotics are valued, and government…
Natural immunity to COVID-19 may be long-lasting
Until now, we didn’t know how long immunity after infection with COVID-19 would last – new research suggests it could be long-lasting.
Coronavirus vaccine results are pouring in, and it's good news for older people
Early results from COVID-19 vaccine trials are starting to emerge, and scientists have received the first reports from three independent studies with optimism because protection against the coronavirus is possible.
Rediscovering the public health origins of WASH
As COVID-19 continues to surge over the globe, it forces us to reckon with the fact that true global health equity – universal access to primary healthcare – is an unfinished task that we pay for at our collective expense. Water, sanitation…
Moderna follows Pfizer with exciting vaccine news – how to read these dramatic developments
It is very exciting to hear another positive story about vaccine trial results – a good vaccine is the most likely way of ending the pandemic.
The Human Cost of COVID-19: Eugene
Vienna, Austria – In this final story in the World Health Organization series exploring the human cost of the COVID-19 pandemic, Eugene, a care-home nurse in Austria, shares the story of how he survived coronavirus.
Volunteers take lead on contact tracing in hard-to-reach communities
As COVID-19 has overwhelmed the medical system, a group of volunteers in Nigeria is undertaking the task of slowing the disease’s spread within communities.
Vaccine roll-out is still months away – how can we avoid more lockdowns in the meantime?
The main way the virus is thought to spread is through respiratory droplets and fomites (contaminated objects or materials such as clothes, furniture and door handles). With this mode of spread, most infections take place through close contact,…
5 ways COVID-19 is exacerbating global poverty
COVID-19 has presented many new social and economic challenges, and is exacerbating already existing ones. One such challenge is global poverty. Right now over 700 million people live in extreme poverty worldwide, which is defined as living on…
The Human Cost of COVID-19: Gracia
In this third story in the World Health Organization series exploring the human cost of the COVID-19 pandemic, Gracia, a junior high school student in West Papua, learns life skills for the future.
'Back to normal by spring': are we expecting too much from the first COVID-19 vaccines?
Pfizer’s update certainly is fantastic news. A COVID-19 vaccine could well be approved and ready for use in the next few months. But whether that means we can all get back to normal life by early 2021 is less certain.
Until a coronavirus vaccine is ready, pneumonia vaccines may reduce deaths from COVID-19
The yearly influenza season threatens to make the COVID-19 pandemic doubly deadly, but I believe that this isn’t inevitable.
Coronavirus: believing in conspiracies goes hand in hand with vaccine hesitancy
While developing an effective vaccine probably won’t bring an immediate end to the pandemic, it’s clear that things can’t begin to return to normal without one. Anything that reduces a future vaccine’s effectiveness will be a problem. This…
Come together: how the fight to end polio can help the COVID-19 recovery
The world is anxiously awaiting the development of a safe and effective COVID-19 vaccine. Nearly 60 years ago, the world was also waiting in eager anticipation of a publicly available vaccine to stop a disease that was ravaging communities around…
How deliberately infecting volunteers with COVID-19 could help accelerate vaccine development
Josh Morrison, Co-founder and Executive Director of 1Day Sooner, an organisation that advocates on behalf of challenge trial volunteers, explains why.
Equitable COVID-19 vaccine distribution will lead to the biggest reduction in deaths
Mathematical modelling suggests that if wealthy countries stockpile COVID-19 vaccines, we will see nearly twice as many deaths than if vaccines were shared equally across the globe.
Overcoming the COVID-19 Disruption to Essential Health Services
It is clear that COVID-19 will persist much longer than anticipated. If countries do not take action soon to ensure the continuity of essential health services during the pandemic, the future death toll from communicable and noncommunicable…
Digitizing vaccine cold chain, key to post-pandemic immunization in Indonesia
For some vaccinators in the vast archipelagic nation of Indonesia, a day in their professional life may involve trekking through deep tropical valleys and jungles or crossing the open waters on rickety boats to isolated islands.
Why protecting, promoting essential services for women and children is now more critical than ever
Now, COVID-19 is unleashing substantial health, social and economic impacts in every corner of the globe. But it is the poorest countries and the most vulnerable communities around the world that stand to suffer the most from this protracted…
Let’s flatten the infodemic curve
We are all being exposed to a huge amount of COVID-19 information on a daily basis, and not all of it is reliable. Here are some tips for telling the difference and stopping the spread of misinformation.
Ghana launches polio campaigns despite the challenges of COVID-19
In the face of the challenges posed by the pandemic, Ghana is working to protect 4.5 million at-risk children from polio through the launch of two polio vaccination campaigns. Here’s a closer look at those efforts for World Polio Day.
The COVID-19 pandemic has shone a spotlight on social, economic, and environmental risks that have been building for the past half-century of neoliberalism. Even amid the deep uncertainties of today's global situation, one thing is clear: it is…
We need a global response to the COVID-19 pandemic
President of Niger H.E. Issoufou Mahamadou talks about the importance of routine immunisation and Gavi support in Niger, and the need for ensuring universal access to COVID-19 vaccines through the COVAX Facility.
Timor-Leste’s Director-General of Health Services for the Ministry of Health discusses the unique challenges of tackling COVID-19 in Timor-Leste and how have they been addressed.
Why we need a “portfolio approach” to COVID-19 vaccine development
Most vaccine candidates in early development fail: this is the stark reality of vaccine development.
Children who get zero vaccines deserve 100% of our attention
Over 50% more children were immunised in Gavi countries in 2019 than in 2000, yet 10.6 million children continue to miss out entirely on basic vaccinations. Here’s why reaching these children and setting them on the pathway to full immunisation…
Once we have effective Covid-19 treatments, it shouldn’t only be the rich who benefit
We need a range of treatments to make Covid-19 preventable and treatable. Jeremy Farrar describes recent progress made by research and why more investment is needed.
Brazil city ‘might have reached herd immunity'
Herd immunity threshold may have been reached in Amazonas capital, Manaus, but experts warn that herd immunity is not the end of the pandemic.
COVID-19 as an awakening for hand hygiene access
Globally 3 billion people lack hand hygiene facilities at home and two out of five health care facilities lack hand hygiene at points of care. As the pandemic unfolded around the world, hand sanitizer and handwashing stations became as in-demand…
Strong systems for strong vaccine coverage
In 2019, countries delivered more than 230 million routine vaccinations, a record number, supported by unprecedented levels of health system funding from Gavi to support immunisation and protect the most vulnerable children. Here is what Gavi did…
What is the world like for girls today?
On International Day of the Girl Child, Gavi’s Megan Holloway looks at how COVID-19 is impacting the lives of girls.
A Moment of Truth in the Pandemic
As the most ambitious pandemic-response initiative ever conceived, the COVID-19 Vaccine Global Access Facility is the best chance the world has to bring the pandemic to an end. But to succeed, COVAX requires broad international buy-in, based on…
Immunization continues amid COVID-19
European Commission Civil Protection and Humanitarian Aid Operations (ECHO) humanitarian air bridge made it possible.
Protecting community health workers means protecting communities in the DRC
Preventing and controlling infectious disease is possible with dedication of health workers, multi-stakeholder collaboration and equitable access to vaccines. Africa’s wild polio-free certification, which took decades of collaborative effort, is…
AVADAR: How digital health fast-tracked Nigeria’s drive to eradicate polio
Polio, a contagious disease caused by the poliomyelitis virus is transmitted primarily when stool from someone infected contaminates foods, drinks or water sources. It invades the nervous system and could, in a matter of hours, lead to complete…
Why I volunteered for a COVID-19 vaccine trial
Vaccines are designed to trigger an immune response to pathogens we’ve never encountered before. But how does it feel to be one of the first humans to encounter a new vaccine? British communications consultant Heather Macdonald-Tait explains what…
How COVID-19 is heightening the economic risks in Gavi-supported countries
In a matter of months, the COVID-19 pandemic has devastated the global economy. Now a new study looks at how this economic shock is impacting sovereign debt levels of Gavi-supported countries.
Q&A with Professor Salim Karim
Africa has not been as hard hit by the COVID-19 pandemic as predicted. Here, we talk to Professor Salim Abdool Karim, the head of South Africa's Ministerial Advisory Committee on COVID-19 to find out why and what the future might hold.
Understanding urban challenges in Uganda
In the first of this series, we look at the highlights of an evaluation to determine the potential barriers to immunisation caused by the increase in urbanisation.
Why the world is at risk without immunisation
Vaccines save lives by protecting children and adults from diseases. COVID-19 pandemic is a clear example of what a world without vaccines could look like. Governments should ensure that delivery of essential services such as vaccines remains a…
How good quality data is vital to saving lives
Throughout the 2016–2020 strategic period, the Vaccine Alliance has worked with countries to improve data quality. From GIS to SMS, new tech and tools aim to reach more people with life-saving immunisation.
How Gavi’s co-financing model works
To bring countries on a trajectory towards financial sustainability, and to empower them to take ownership of their vaccination programmes, Gavi has pioneered an approach to co-financing and transition.
A Nurse in Uganda is a Trusted Messenger of Hope
Her own healthy baby son is how Ugandan nurse Maureen Wandawa demonstrates the benefits of immunization. Working together to build confidence in, and access to, vaccines for all who need them is one way we can #UniteforHealth to keep us all safer…
Download for life: a mobile app to improve vaccine coverage in Côte d’Ivoire
Gavi's partnership with telecoms provider Orange and the Ministry of Health overcomes barriers to childhood immunisation through tailored, tested technology.
More than one million people have died of COVID-19
The true number could be much higher, and two million could die before we get a vaccine, says the World Health Organization.
Recovering Better through Gender Equity
Gender equality must be at the heart of not only our response, but our attempts to restructure systems to be fairer and more equal to recover better.
Global investors must support pharma solidarity and collaboration in the response to COVID-19
The prospect of having the first supplies of at least a few effective COVID-19 medicines and vaccines by the end of 2020 is a key driver of recent market optimism.
The benefits of immunisation extend beyond preventing childhood deaths. Vaccines are also a vital tool in the fights against cancer, outbreaks and antimicrobial resistance.
The world needs Gavi now more than ever
When I join Gavi in January, it will be at a time when it is contemplating the most ambitious period in its history.
The last mile of polio eradication and the vaccination challenges it brings
We are close to a polio-free world, yet cases of vaccine-derived disease are challenging the eradication effort.
The goal of our market shaping work is to help ensure vaccine markets work better for lower-income countries. We want the maximum number of people to receive the life-saving and health-protecting benefits of immunisation.
Experimental approaches to address post-transition risks
After transitioning out of Gavi support in 2017, the Republic of Moldova is mobilising champions and advocates to strengthen vaccine confidence – which is crucial to the success and sustainability of immunisation programmes.
Keep focus on emerging infections, Disease X: analysts
Biggest funders of emerging infectious diseases R&D are also biggest recipients. Ebola and Zika experienced R&D spike in wake of epidemics. US is losing global leadership role in the field.
How to keep vaccine production going during a pandemic?
Vaccine manufacturers have worked hard to adapt to a new normal of keeping supplies for immunisation programmes going in the midst of the COVID-19 pandemic.
Typhoid: immunising against antimicrobial resistance
Typhoid fever is increasingly resistant to the main antibiotics used to treat it. Gavi is supporting a new vaccine to help control this deadly disease – and fight antimicrobial resistance.
Gavi at UNGA: Global solidarity to defeat COVID-19 and create a better world for all
September 2020 marks the 75th anniversary of the United Nations General Assembly (UNGA). This year, the focus of the high-level General Debate is the importance of effective multilateralism.
What does the pausing of the AstraZeneca trial mean for other COVID-19 vaccines?
Gavi’s Derrick Sim, Director of Vaccine Supply and Demand, discusses why it is sometimes important to halt clinical trials.
Leaders in the fight against infectious diseases
Snapshots from DRC, India and Zambia illustrate country ownership and progress in the fights against Ebola, polio and HPV.
10 reasons why pandemic fatigue could threaten global health in 2021
Being in a constant state of high alert and uncertainty is exhausting yet health officials warn that we can’t let our guard down yet and resisting living with the ‘new normal’ could threaten our health.
Coronavirus vaccine: why it’s important to know what’s in the placebo
Some researchers conducting clinical trials on a COVID-19 vaccine have not revealed to the public what the placebo contains, but they should.
How health systems work and why they matter
Supporting health systems is essential for Gavi’s work to improve immunisation coverage and equity.
Could COVID-19 be fuelling drug resistance?
We need to understand the impact of Covid-19 on wider health issues to shape better public health responses and limit long-term consequences. Drug resistance is one of these, Gemma Buckland-Merrett explains.
Resilience and the risks to global health security
When disaster strikes a country, the impact on people’s health can be immense and long-lasting. As the effects of climate change are felt around the world through rising temperatures and sea levels, natural disasters are increasingly frequent and…
When I was 12, I received the HPV vaccine just like most girls my age in Rwanda. The day after we got the first dose, my classmates started spreading rumours about the vaccine that they had heard from their families and communities.
Vaccines are the best tools yet invented to prevent child deaths and protect people’s health. Gavi funding for access to vaccines has protected a generation of children growing up in lower-income countries. Gavi funds twin approaches to…
The three Vs needed to end this pandemic
While scientific and vaccine manufacturing community make huge strides in the race towards that goal, it is important to remember that vaccines are only one of three Vs needed to beat this coronavirus.
The role of social mobilisers during COVID-19
How one social mobiliser in South Sudan has managed to contribute to health security in her region.
Our shared future is not a choice
2020 was already going to be a monumental year — the beginning of the 10-year countdown for achieving the Sustainable Development Goals (SDGs).
Mapping vaccine confidence worldwide
Anecdotes of how people feel about vaccines are plenty, but there have been few attempts to gather robust global data. This five-year study reviewed survey responses from over a quarter of a million people around the world on how they felt about…
A record-breaking year for childhood immunisation
Gavi’s CEO provides an overview of its flagship publication, launched today.
Malawi: Reaching girls earlier
In Malawi, primary school drop-out rates are among the highest in Africa. To try and guarantee high coverage, the HPV vaccine is administered to 9-years-old girls.
COVID-19 vaccines: what happens after clinical trials
If a COVID-19 vaccine is to reach the global public sometime next year, it will be the result of one of the fastest developments and rollouts of a vaccine against a new disease ever. In contrast, the RTS,S malaria vaccine, which is currently…
How Ethiopia is maintaining health services during a pandemic?
Ethiopia has overtaken Nigeria to become the Gavi-eligible African country most affected by COVID-19. Over the past week, the number of confirmed cases in Ethiopia has grown exponentially. Despite the pandemic, immunisation activities – both…
COVID vaccine is being speeded up but urgency cannot be at expense of safety, says Dr Seth Berkley
As CEO of Gavi, the Vaccine Alliance funded largely by Bill Gates, Dr Seth Berkley has helped vaccinate almost half the world’s children from fatal and debilitating diseases. His current baby is the COVAX Facility to help develop, manufacture and…
Supply chains: The lifeline for getting PPE to community health workers
You may have heard the saying, “no products, no programs,” but for those who work in public health during COVID-19, the saying has become “no PPE, no programs.” And for community health workers (CHWs) who play a vital role in providing health…
Ethiopia Reaching girls at scale
Helping rural and urban girls across Ethiopia understand cervical cancer and HPV.
Modelling the Manufacturing Process for COVID-19 Vaccines: Our Approach
Researchers across the world are working flat out to develop and manufacture a vaccine for COVID-19 that can end what has been the worst pandemic in at least a century.
How to measure the success of a COVID-19 vaccine?
Dr Melanie Saville, CEPI’s Director of Vaccine Development, discusses what a successful vaccine against COVID-19 would involve, some of the unknowns and the challenges that vaccine developers are wrestling with, and what makes CEPI’s approach…
Before the pandemic began, the Democratic Republic of the Congo (DRC) was already facing a severe measles outbreak which started at the end of 2018. Despite huge challenges caused by COVID-19 and other diseases, the largest measles outbreak in…
How the African Region is Celebrating the End of Wild Polio while cVDPV Outbreaks Continue
DRC: The great lengths that polio vaccinators go to reach every last child
By foot, boat and bicycle, thousands of volunteer vaccinators in the Democratic Republic of Congo (DRC) go to extraordinary efforts, sometimes at personal risk, to help eradicate polio.
An estimated $15.9 trillion has been mobilised to respond to the health and economic impact of the COVID-19 pandemic. Some of this has gone to multilateral and bilateral funders to support low- and middle-income countries’ (LMIC) governments.
Borno: Africa’s Last Wild Poliovirus Frontier
When Aisha took her son Busami Modu to his grandmother’s house in Kuya, a village in Borno state, Nigeria, she waved goodbye to a healthy, happy little boy. A “beauty to behold,” she recalls. The next time she saw him, he was paralysed and unable…
Data from global Ipsos survey shows 37% of those polled worldwide think it is likely a vaccine will be available by the end of the year. But is this optimism misplaced?
How is Nigeria addressing the public health challenges due to COVID-19?
As one of the African countries most affected by COVID-19, Nigeria has faced significant economic and health impacts, including to routine immunisation, in the face of the pandemic.
What do diarrheal disease and COVID-19 have in common?
Diarrheal disease is the second leading infectious killer of children under five worldwide. Lessons learned from tackling this deadly disease could also help us recover from COVID-19 stronger than ever.
How has our urban world made pandemics more likely?
As more people have moved to cities, population density, human encroachment and increased global interconnectivity have contributed to the spread of infectious diseases.
COVID-19 has triggered lockdown measures for billions of people around the world. As many of us struggle to adapt to the ‘new normal’, we look at the origins of quarantine measures - and how they’ve been used to contain deadly outbreaks for…
How creative communication strategies are helping fight COVID-19 misinformation in DRC
The misinformation surrounding COVID-19 in the Democratic Repubic of the Congo isn’t new – public health officials have seen rumours and myths circulate with Ebola. Here’s how they are tackling them.
New evidence shows investments in vaccination produce even greater returns than previously thought
Analysis suggests that investments in vaccination programmes will result in US$ 0.8 trillion in returns over the next ten years.
You’ve got your antibody test result – but what does it mean?
If you test positive for COVID-19 antibodies, does that actually mean you’ve been infected?
Children missing out on routine vaccinations in Somalia amid COVID-19 fears
In the recent past, 25-year-old Zahra Mohamud* had visited the Hawadle maternal and child health centre in Hargeisa regularly to have her older son Ibrahim* vaccinated against the most common childhood diseases.
Now is the Time to Pave the Way for Equitable Vaccine Distribution
When COVID-19 vaccines become available, one of the biggest challenges the global community will face is equitable distribution. This will be the single largest vaccine deployment in history.
How are Gavi-supported countries maintaining routine immunisation during the pandemic?
A spotlight on Ethiopia, India, the Solomon Islands and Zambia
Governments have had to act fast to not only protect their people against COVID-19 but also to ensure protective measures like vaccination against other infectious diseases…
Why the UK wants to recruit half a million people in a COVID-19 vaccine trial
The UK has just secured 90 million doses of future COVID-19 vaccines, and now it is trying to recruit volunteers for some of the biggest ever phase 3 trials to test potential vaccines.
Delivering life-saving vaccines during the COVID-19 pandemic
Health workers are stopping at nothing to ensure immunizations for every child.
What happens if COVID-19 mutates?
Reports of a potentially more infectious form of the coronavirus have fed fears that a "mutant” version of the virus could arise. But what does mutation actually mean and is it really a cause for concern?
Should countries stop vaccinations to halt the spread of COVID-19?
It’s a devil’s choice governments have faced since the start of the pandemic, but new research could provide the answer.
Could COVID-19 be airborne, and if so how do we protect ourselves?
Ever since the new coronavirus was identified, there has been a debate about whether it is airborne or not. Now, over 200 scientists from around the world have written to the World Health Organization urging it to consider the potential of COVID-…
The vaccines success story gives us hope for the future
As the world waits for a vaccine to defeat the COVID-19 pandemic, we look back to all that vaccines have achieved for humanity.
Q&A with Ola Rosling of Gapminder
The head of Gapminder, whose mission is to fight ignorance with a fact-based world view, talks with Gavi about data and COVID-19.
How Technology Helps Health Workers Deliver Essential Care in Developing Countries
Living Goods saves lives by supporting digitally empowered community health workers who deliver care
This article is a summary of an online conversation hosted by the Gavi CSO Constituency in partnership with ACTION, Gavi, the Vaccine Alliance, RESULTS UK & Save the Children.
How the COVID-19 lockdown is affecting routine immunisation
Routine immunisation has slowed in many Gavi-supported countries around the world. Here, researchers writing in The Lancet, including a Gavi scientist, look at the effect of lockdown on vaccination programmes in Karachi, Pakistan …
30 June 2020: Overview of COVID-19 situation in Gavi-supported countries and Gavi’s response
Across Gavi-eligible countries, we have seen a consistent increase in cases since March 2020. Within the month of June alone, the number of cases has more than doubled, with 70 out of 73 Gavi-eligible countries reporting over 1 million confirmed…
How is the pandemic impacting one of the world’s largest refugee settlements?
As of 27 June 2020, there have been 513 tests carried out in the camps, with 49 confirmed cases.
First African trial of a COVID-19 vaccine
The first African clinical trial for a COVID-19 vaccine has started in South Africa, run by the University of the Witwatersrand, Johannesburg (Wits).
'Global Goal: Unite for Our Future' mobilized over $6.9 billion to help the world’s most vulnerable.
What is the COVAX pillar, why do we need it and how will it work?
With more than 200 COVID-19 vaccines already in development, why do we need the Access to COVID-19 Tools Accelerator Vaccine Pillar, or COVAX Pillar?
Seven things countries have done right in the fight against COVID-19
There has been a huge variation in how countries have responded to COVID-19. Whilst some have imposed strict lockdown measures, others have avoided tight restrictions. But what works best?
16 June 2020: Overview of the COVID-19 situation in Gavi-supported countries and Gavi’s response
Seventy (of 73) Gavi-eligible countries have reported 824,259 confirmed cases and 20,641 deaths. The number of cases is increasing at an average rate of 4% from the previous day.
As the devastating consequences of the pandemic unfold across the world, COVID-19 is making long-lasting changes in global health
Is COVID-19 about to take off in Africa?
So far relatively few people in Africa have been infected by the novel coronavirus, with around 215,000 cases and 5,800 deaths in a population of 1.2 billion in the continent. But with lockdowns lifting, will cases soar?
Vaccinations and COVID-19: What parents need to know
How to safely get routine vaccinations for your child during the COVID-19 pandemic
5 reasons why pandemics like COVID-19 are becoming more likely
SARS, Ebola, Zika, and now COVID-19. Five reasons why disease epidemics and even pandemics are becoming increasingly common.
Could COVID-19 ever be eradicated?
Ideally, humanity would end COVID-19 by eradicating it so there are no new cases globally even in the absence of preventive measures. However, experience with other disease eradication programmes indicates that this will be very challenging, and…
How the world changed on 4 June
On 4 June, global leaders met at a virtual event to pledge support for Gavi, the Vaccine Alliance, and in the process made history by reaching new global health milestones
02 June 2020: Overview of the COVID-19 situation in Gavi-supported countries and Gavi’s response
Seventy (out of 73) Gavi-eligible countries have reported 475,401 confirmed cases and 12,464 deaths. Though this number still accounts for a small proportion, about 7%, of global COVID-19 cases, this share is expected to increase given the…
Supporting Gavi now to build a better future
We’ve all seen the shocking projections: at least 80 million children under one are at risk of developing diseases like measles, diphtheria, and polio due to disruptions in routine immunization services because of COVID-19.
How physical distancing, masks and eye protection reduces the spread of COVID-19
In the absence of a vaccine or treatment against the new coronavirus, measures to control its spread rely on avoiding contact with people who we are not sharing a house with. This study shows the varying effects of such preventive measures.
Q&A with Keller C. Rinaudo, CEO and Co-Founder of Zipline
Zipline is a California-based automated logistics company that uses autonomous drones to make on-demand, emergency deliveries of high priority products, including emergency and routine vaccines and other health products.
Immunisation and COVID-19 at the 73rd World Health Assembly
Equitable access to COVID-19 vaccines was a major priority for world leaders attending the virtual meeting
Vaccines for the happy few = health for none
Health is not just a human right, it is a prerequisite for development. Through its advocacy programme Global Health, Global Access, Cordaid continually stresses the importance of Health for All.
We are working hard to ensure that immunization services continue to be available for every child
There is no fundamental reason to stop immunization in any situation.
19 May 2020: Overview of the COVID-19 situation in Gavi-supported countries and Gavi’s response
Seventy (out of 73) Gavi-eligible countries have reported 257,996 confirmed cases and 7,547 deaths. These 70 countries account for a small proportion, about 5%, of global COVID-19 cases and 2% of global deaths.
Here are four ways Vietnam has managed to control COVID-19
Vietnam became one of the first countries to report a COVID-19 infection and the first to report human-to-human transmission outside of China. One hundred days later, just 270 cases had been confirmed in the country, with no deaths.
Experts are warning that COVID-19 could become an endemic disease. What does this mean and how will it impact our response?
How do you keep pregnant women safe during a pandemic?
Pregnant women can be more exposed to risk during outbreaks. With 200 million pregnant women in the world, is the global pandemic response adequate to protect them, and not just from the coronavirus?
Rwanda’s efforts to strengthen its health system paying off in midst of COVID-19 pandemic
The country has made strategic investments including supply chain strengthening and technological innovation, all of which have allowed critical immunisation activities to continue during and beyond the quarantine.
Is air travel risky in the age of COVID-19?
Does air travel increase your chances of getting becoming infected by the novel coronavirus and contracting COVID-19?
Q&A with Jonathan Stambolis, CEO of Zenysis Technologies
Zenysis is an advanced analytics software company. Their platform helps countries harness the life-saving power of data to improve the delivery of essential health services, like childhood vaccination, and to fight infectious disease outbreaks.…
Maintaining commitment to immunization in Africa through COVID-19 and beyond
The COVID-19 pandemic is putting a strain on health systems worldwide. The increasing demand on health facilities and health care workers has dramatically shifted resources from essential routine health services to COVID-19 response as countries…
The poorest are most likely to die from COVID-19
Social inequity will put the poorest populations at greater risk from COVID-19
How can fragile countries, like Afghanistan, respond to COVID-19?
As countries rich and poor struggle with challenges in the face of the COVID-19 pandemic, Afghanistan is taking proactive steps to overcome its own.
How COVID-19 is causing a ‘hunger pandemic’
The new coronavirus has killed more than a quarter of a million people in just a few months, but the pandemic’s effect on food, housing and job insecurity will last years.
Is antimicrobial resistance exacerbating the COVID-19 pandemic?
Will efforts to save lives in cases of severe COVID-19 trigger another global health crisis once the pandemic is over?
5 May 2020: Overview of the COVID-19 situation in Gavi-supported countries and Gavi’s response
Sixty-nine (out of 73) Gavi-eligible countries have reported 123,129 confirmed cases and 3,941 deaths, five more countries than the 64 announced last week. These countries account for about 3% of global COVID-19 cases and deaths.
Five ways coronavirus could impact the future of global health
For the global health community, the implications resulting from COVID-19 could be immense.
28 April 2020: Overview of the COVID-19 situation in Gavi-supported countries and Gavi’s response
The COVID-19 pandemic has placed a heavy burden on global economies, health care systems and other core sectors. Gavi is helping countries reallocate their health system strengthening grants to provide fast and flexible funding for vital…
Keeping routine immunisation going during COVID-19 in Indonesia
Q&A with Dr Vertando Halim, Coordinator of Expanded Programme on Immunization, Jakarta, Indonesia
A COVID-19 vaccine might be ready within 18 months. But what happens then?
The world is currently on pause, buying time while an effective vaccine against COVID-19 is developed. Already, several vaccines have entered human trials, and discussions have begun about how the most promising candidates might be manufactured…
Gavi’s role in the COVID-19 response in supported countries
The COVID-19 pandemic has placed a heavy burden on global economies, healthcare systems and other core sectors. Gavi is helping countries reallocate their health system strengthening grants to provide fast and flexible funding for vital resources…
Lessons in preparing to distribute COVID-19 vaccine
How the 2009 H1N1 influenza pandemic can help us be better prepared for distributing and administering potential COVID-19 vaccines.
COVID-19 threatens weak health systems in indigenous Amazonian communities
Indigenous groups face unique health challenges. How do we mitigate these risks in our COVID response?
Why a gender lens is needed for the COVID-19 response
The immediate and long-term consequences of the coronavirus pandemic are disproportionately impacting the lives of women and girls, and the most marginalised.
Informal workers at risk from both COVID-19 and lockdown measures
With COVID-19 spreading in low- and middle-income countries, governments have been imposing strict lockdowns that could pose an even greater threat than the virus itself.
COVID-19 lessons from a frontline health worker
Interview with Emmanuel Lansana, outpatient supervisor at Monrovia’s Redemption Hospital in Liberia, about lessons learned from the Ebola outbreak that can help tackle COVID-19.
How Africa has developed its scientific research capabilities
Africa is better prepared now to perform research in emerging infections than during the Ebola epidemics in West Africa.
Should we wear masks against COVID-19?
Will new evidence suggesting that the virus is airborne mean we all need to wear masks?
Coronavirus risks creating a lethal vaccine gap
Could the impact of COVID-19 on routine immunisation be an even bigger threat in low-income countries than the virus itself?
Modelling suggests suppression strategy will save more lives from COVID-19 in poor countries
Imperial model of the spread of COVID-19 implies a suppression strategy could be most effective.
How do we stop the spread of a pandemic in a slum?
Over 1 billion lives are at risk if COVID-19 sweeps through crowded slums. How can we prepare?
When a COVID-19 vaccine is available, how will we pay for it?
When a COVID-19 vaccine is available, how will we pay for it?
With zero cases of COVID-19 in Mozambique so far, Dr Rosa Marlene, National Director of Public Health of Mozambique, discusses a potential impact that a pandemic of this magnitude could have on the country’s health system.
How might COVID-19 play out in Africa?
Africa marks a critical juncture for the coronavirus pandemic.
What is an emergency vaccine stockpile and how can they prevent pandemics?
We don’t yet know how the COVID-19 pandemic will progress and how long it will take for a vaccine to be developed, but the knowledge and experience gained from past outbreaks can be applied to the global response to coronavirus.
Will coronavirus herald a new era in vaccine innovation?
A vaccine against the novel coronavirus is under urgent development as the number of people infected with COVID-19 increases, a process which could also result in a new range of highly innovative vaccine technologies that ushers in a new era in…
More from Priya Joi
Recommended for you